Biomarker Validation with Triple Quadrupole Mass Spectrometry: A Complete Guide from Discovery to Clinical Application
This article provides a comprehensive resource for researchers and drug development professionals on the application of triple quadrupole (QqQ) mass spectrometry in biomarker validation.
Biomarker Validation with Triple Quadrupole Mass Spectrometry: A Complete Guide from Discovery to Clinical Application
Abstract
This article provides a comprehensive resource for researchers and drug development professionals on the application of triple quadrupole (QqQ) mass spectrometry in biomarker validation. It covers foundational principles of QqQ instrumentation and Selected Reaction Monitoring (SRM), explores methodological workflows for developing robust quantitative assays, addresses critical troubleshooting and optimization strategies to enhance sensitivity and specificity, and examines validation frameworks and comparative analyses with alternative platforms. The content synthesizes current trends, including the growing adoption of QqQ in clinical applications and its role in translating biomarker candidates into validated clinical tests.
The Triple Quadrupole Foundation: Core Principles and Its Central Role in the Biomarker Pipeline
Core Components and Operating Principle
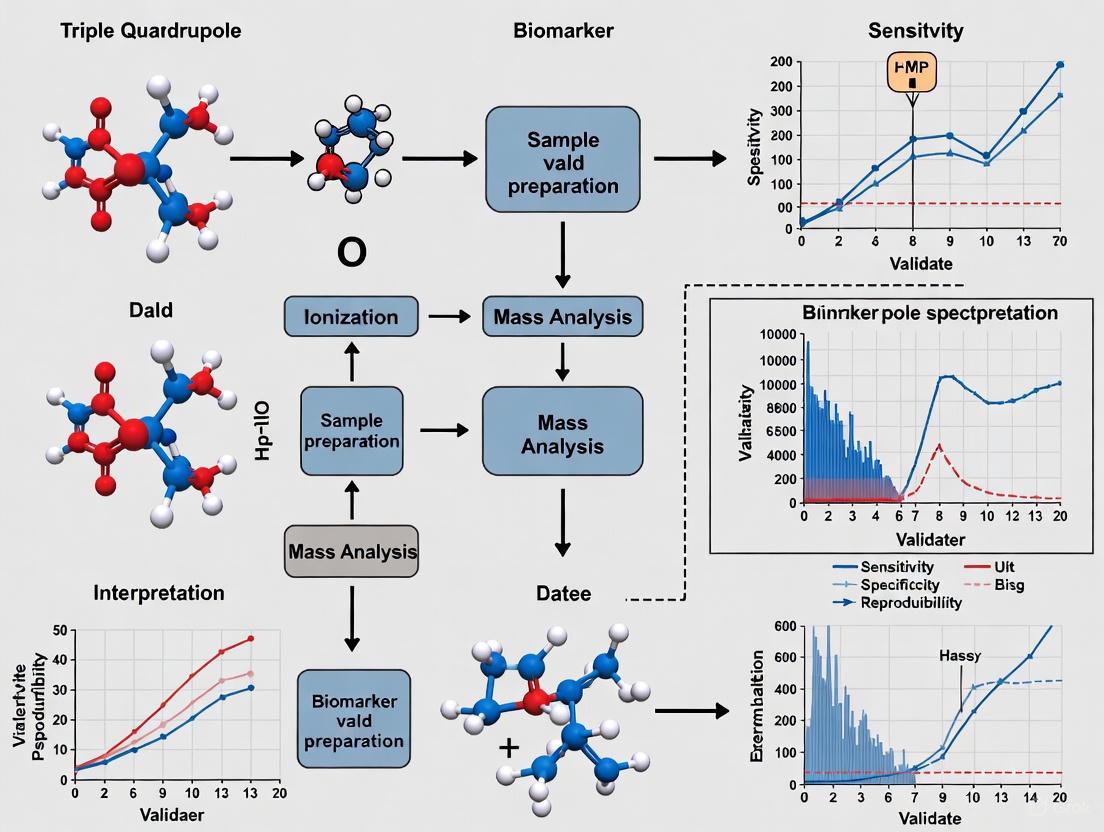
The triple quadrupole mass spectrometer (TQMS), often denoted as the QqQ configuration, is a tandem mass spectrometer consisting of two mass-resolving quadrupoles (Q1 and Q3) separated by a non-mass-resolving radio frequency (RF)–only quadrupole that acts as a collision cell (q2 or Q2) [1] [2]. This instrumental setup operates on the principle of tandem-in-space mass spectrometry, where different mass-selective processes occur sequentially in separate physical regions of the instrument [1].
The fundamental operation involves ionization of the sample, primary mass selection in Q1, collision-induced dissociation (CID) in Q2, mass analysis of the resulting fragments in Q3, and finally, detection [1]. The system's robustness, sensitivity, and quantitative accuracy have cemented its role as a cornerstone technology in modern analytical chemistry, particularly in biomedical research and clinical applications where its use has increased 2–3 times this decade [3] [4].
Function of Individual Quadrupoles
Q1: The First Mass Filter The first quadrupole (Q1) serves as the primary mass-to-charge (m/z) selector. It consists of four parallel, cylindrical metal rods controlled by a superposition of direct current (DC) and radio-frequency (RF) voltages [1] [2]. By precisely varying these RF and DC potentials, Q1 can be tuned to allow only ions of a single, specific m/z value to pass through to the next stage while rejecting all others [2] [5]. This selective transmission provides the first stage of mass filtration, isolating the precursor ion of interest for subsequent fragmentation and analysis.
Q2: The Collision Cell The second quadrupole (Q2 or q) functions as a collision cell and is typically operated with only an RF voltage, meaning it does not perform mass resolution [1] [2]. Its primary role is to induce fragmentation of the precursor ions selected by Q1. This is achieved through collision-induced dissociation (CID), where the precursor ions collide with an inert gas such as argon, nitrogen, or helium [1] [5]. These collisions convert kinetic energy into internal energy, causing the precursor ions to break apart into characteristic fragment or product ions [2]. In some modern instruments, the normal quadrupole collision cell has been replaced by hexapole or octopole cells to improve ion transmission efficiency [1].
Q3: The Second Mass Filter The third quadrupole (Q3) is the second mass-resolving analyzer, structurally identical to Q1 and also controlled by DC and RF potentials [1] [2]. It receives the product ions generated in Q2 and performs the final stage of mass analysis. Depending on the analytical mode, Q3 can be set to scan the entire m/z range of the fragments, monitor for a specific product ion, or correlate its scanning with Q1 [2]. This second stage of mass filtration is key to the exceptional selectivity of the triple quadrupole.
Table: Core Functions of the Triple Quadrupole Components
| Component | Common Designation | Primary Function | Key Operational Characteristic |
|---|---|---|---|
| First Quadrupole | Q1 | Primary mass selection; filters precursor ions | DC and RF voltages filter by m/z [1] [2] |
| Second Quadrupole | Q2 or q | Collision cell; fragments precursor ions | RF-only field; contains collision gas (e.g., N₂, Ar) [1] [5] |
| Third Quadrupole | Q3 | Secondary mass analysis; filters product ions | DC and RF voltages filter fragment ions by m/z [1] [2] |
Operational Scan Modes
The power and versatility of the triple quadrupole mass spectrometer are realized through its diverse scan modes. By independently controlling the mass-selective functions of Q1 and Q3, the instrument can be configured for various experiments tailored to qualitative identification or highly sensitive quantification [1] [2]. The most common scan modes include Product Ion Scan, Precursor Ion Scan, Neutral Loss Scan, and Selected/Multiple Reaction Monitoring.
Triple Quadrupole Operational Flow
Key Analytical Modes
Product Ion Scan In this mode, Q1 is set to select a specific precursor ion of a known mass, which is then fragmented in Q2. Q3 is set to scan across a range of m/z values to record all the resulting product ions. This scan provides a characteristic fragmentation pattern, which is invaluable for structural elucidation of the original ion and is commonly used to identify optimal ion transitions for quantitative methods [1] [5].
Precursor Ion Scan A precursor ion scan involves setting Q3 to monitor for a specific product ion that is characteristic of a particular functional group or molecular moiety. Q1 then scans a wide m/z range. Any precursor ion that fragments to produce the characteristic product ion monitored in Q3 is identified. This mode is highly selective for recognizing ions sharing a common structural feature in a complex mixture [1].
Neutral Loss Scan In a neutral loss scan, both Q1 and Q3 are scanned simultaneously but with a constant mass offset between them. This configuration selectively detects all ions that lose a specific neutral fragment (e.g., H₂O, NH₃, CO₂) during fragmentation in Q2. Like the precursor ion scan, this mode is useful for selectively identifying closely related compounds within a sample [1] [5].
Selected Reaction Monitoring (SRM) / Multiple Reaction Monitoring (MRM) This is the most widely used mode for high-sensitivity quantification. In SRM/MRM, both Q1 and Q3 are fixed at specific m/z values to monitor a single, defined transition from a specific precursor ion to a specific product ion [1] [2]. This dual mass-filtering process provides exceptional selectivity and dramatically reduces chemical noise, leading to significantly lower limits of detection and quantification. When multiple such transitions are monitored in a single run, it is termed Multiple Reaction Monitoring (MRM), enabling high-throughput, multiplexed quantification of dozens of analytes simultaneously [1] [2]. The specificity is often enhanced by monitoring two mass transitions for a single analyte: a quantifier ion for concentration measurement and a qualifier ion to confirm the analyte's identity [2].
Table: Summary of Triple Quadrupole Scan Modes
| Scan Mode | Q1 Function | Q3 Function | Primary Application |
|---|---|---|---|
| Product Ion Scan | Selects single m/z | Scans a mass range | Structural elucidation, method development [1] [5] |
| Precursor Ion Scan | Scans a mass range | Selects single m/z | Selective detection of ions with a common fragment [1] |
| Neutral Loss Scan | Scans | Scans with fixed offset | Selective detection of ions that lose a common neutral group [1] |
| SRM/MRM | Selects single m/z | Selects single m/z | Highly sensitive and selective target quantification [1] [2] |
Experimental Protocol: MRM-Based Biomarker Quantification
This protocol details the application of a triple quadrupole mass spectrometer coupled with liquid chromatography (LC-MS/MS) for the absolute quantification of a protein biomarker candidate in human plasma using the Multiple Reaction Monitoring (MRM) approach. This workflow is central to biomarker validation studies in drug development [2].
Sample Preparation
- Internal Standard Addition: Add a known quantity of a stable isotope-labeled (SIL) analog of the target protein (e.g., 15N-labeled) to a measured volume of plasma (typically 50-100 µL) [2] [6]. The SIL protein serves as an internal standard to correct for variability in sample processing and ionization.
- Protein Digestion:
- Denature the plasma proteins using a buffer such as ammonium bicarbonate.
- Reduce disulfide bonds with dithiothreitol (DTT) and alkylate with iodoacetamide.
- Digest the proteins into peptides using a sequence-grade protease, most commonly trypsin, overnight at 37°C [2].
- Solid-Phase Extraction (SPE): Desalt and concentrate the resulting peptide mixture using a C18 solid-phase extraction cartridge to remove interfering salts and buffers, eluting peptides with an organic solvent like acetonitrile.
- Reconstitution: Evaporate the eluent to dryness under a gentle stream of nitrogen and reconstitute the peptide pellet in a mobile phase compatible with the LC system (e.g, 0.1% formic acid in water).
LC-MS/MS Analysis
- Liquid Chromatography (LC):
- Column: Use a reverse-phase C18 analytical column (e.g., 2.1 mm x 100 mm, 1.8 µm) for peptide separation [2].
- Mobile Phase: A) 0.1% Formic acid in water; B) 0.1% Formic acid in acetonitrile.
- Gradient: Employ a linear gradient from 2% B to 35% B over 10-20 minutes to elute the peptides based on hydrophobicity [2].
- Mass Spectrometry (MS) - MRM Acquisition:
- Ionization: Utilize Electrospray Ionization (ESI) in positive ion mode [2].
- Source Conditions: Optimize parameters like nebulizing gas flow, drying gas flow, and interface temperature for robust ion generation.
- MRM Transitions: For each target peptide (proteotypic for the biomarker and its SIL analog), program the instrument to monitor at least two specific precursor ion → product ion transitions.
- Quantifier Transition: The most intense transition for calculating concentration.
- Qualifier Transition: A secondary transition to confirm peptide identity based on the intensity ratio relative to the quantifier [2].
- Collision Energies: Optimize the collision energy in Q2 for each specific transition to achieve efficient and reproducible fragmentation.
Data Analysis and Quantification
- Peak Integration: Manually or automatically integrate the chromatographic peaks for the quantifier and qualifier transitions for both the endogenous peptide and the SIL internal standard peptide.
- Analyte Verification: Confirm the identity of the target peptide by ensuring the retention time of the endogenous peptide matches that of the SIL internal standard and that the ratio of qualifier-to-quantifier peak areas is consistent with that of the standard.
- Quantification: Calculate the peak area ratio (Areaendogenous / AreaSIL). Plot this ratio against the known concentration of the internal standard to create a calibration curve. The concentration of the target biomarker in the original plasma sample is determined by interpolating from this curve [2].
The Scientist's Toolkit
Table: Essential Reagents and Materials for LC-MS/MS Biomarker Assay Development
| Item | Function / Explanation |
|---|---|
| Stable Isotope-Labeled (SIL) Protein/Peptide | Serves as an internal standard; corrects for sample preparation losses and ion suppression, enabling absolute quantification [2]. |
| Sequence-Grade Trypsin | High-purity protease for reproducible and complete protein digestion into measurable peptides [2]. |
| Solid-Phase Extraction (SPE) Cartridges (C18) | For sample clean-up, desalting, and concentration of peptides prior to LC-MS/MS analysis, reducing matrix effects [2]. |
| Reverse-Phase LC Columns (C18) | Provides high-resolution separation of complex peptide mixtures, reducing ion suppression and isobaric interferences [2]. |
| Ammonium Bicarbonate Buffer | A volatile buffer suitable for protein digestion and compatible with mass spectrometry, as it can be easily removed during evaporation. |
| Mass Spectrometry Calibrant Solution | A standard solution containing compounds of known mass for regular mass accuracy calibration of the instrument. |
Understanding Selected Reaction Monitoring (SRM) and Multiple Reaction Monitoring (MRM)
Selected Reaction Monitoring (SRM) and Multiple Reaction Monitoring (MRM) are targeted mass spectrometry techniques primarily utilized for the precise quantification of specific analytes within complex mixtures [7]. These methods involve isolating and monitoring predefined precursor-product ion transitions, enabling high selectivity and sensitivity even in challenging biological matrices like serum, plasma, and urine [7] [8]. The fundamental difference between SRM and MRM lies in their monitoring scope: SRM typically involves monitoring a single predefined precursor-to-product ion transition, whereas MRM extends this capability to simultaneously monitor multiple precursor-to-product ion transitions within a single analysis, enhancing throughput and efficiency in quantification experiments [7].
The development of SRM/MRM is rooted in the advancements of tandem mass spectrometry, particularly with the introduction of triple quadrupole mass spectrometers in the late 1970s [7]. These instruments provided the foundational capability to perform targeted quantification by selecting precursor ions in the first quadrupole, fragmenting them in a collision cell, and then selecting specific product ions in the third quadrupole for detection [7] [9]. This technical evolution addressed the growing need for more targeted and quantitative analysis across various fields including pharmaceuticals, clinical diagnostics, and environmental monitoring [7]. In recent decades, SRM/MRM has become indispensable in scientific disciplines such as proteomics, metabolomics, and biomarker validation, with technological advancements further expanding their utility and versatility [7] [10].
Technical Principles and Instrumentation
Fundamental Operating Principles
At its core, SRM/MRM methodology enhances standard mass spectrometry through tandem mass spectrometry (MS/MS) configurations [7]. The process begins with precursor ion selection, where specific precursor ions corresponding to target analytes are chosen based on their unique mass and fragmentation patterns in the first quadrupole (Q1) [7]. These selected ions then undergo controlled fragmentation through collision-induced dissociation (CID) in a collision cell (q2), producing characteristic product ions [7]. The final critical step involves product ion selection, where in SRM, a single product ion corresponding to the expected fragmentation is monitored, while MRM allows simultaneous monitoring of multiple product ions, enhancing throughput and versatility [7]. The specific pair of m/z values associated with the precursor and fragment ions is referred to as a "transition," and the combination of several transitions with the retention time of the targeted peptide can constitute a definitive assay [11].
The instrumentation central to SRM/MRM experiments is the triple quadrupole mass spectrometer, which consists of three quadrupole analyzers arranged in series [7] [9]. The first (Q1) and third (Q3) quadrupoles act as mass filters, while the second quadrupole (q2) serves as a collision cell [9]. This specific configuration enables the non-scanning, static monitoring of predefined transitions, resulting in a significantly increased duty cycle compared to full-scan techniques [11]. The two stages of mass selection with narrow mass windows provide exceptional selectivity, effectively filtering out co-eluting background ions [11]. This technical approach provides a linear response over a wide dynamic range of up to five orders of magnitude and enables the detection of low-abundance proteins in highly complex mixtures, which is crucial for systematic quantitative studies in systems biology and biomarker validation [11].
Comparative Mass Spectrometry Techniques
SRM/MRM offers distinct advantages compared to other mass spectrometry techniques. It provides enhanced selectivity by specifically monitoring predefined precursor-to-product ion transitions, which minimizes interference from background noise and co-eluting compounds [7]. The technique also delivers increased sensitivity by selectively amplifying the signal of target analytes while suppressing background noise, enabling detection even at low concentrations [7]. Furthermore, the ability to precisely control precursor and product ion transitions, coupled with robust calibration strategies, ensures high quantitative accuracy and reproducibility in SRM/MRM analyses [7].
Table: Comparison of Targeted Mass Spectrometry Approaches
| Feature | MRM (SRM) | Parallel Reaction Monitoring (PRM) |
|---|---|---|
| Instrumentation | Triple Quadrupole | Orbitrap, Q-TOF |
| Resolution | Unit resolution | High (HRAM) |
| Fragment Ion Monitoring | Predefined transitions | All fragments (full MS/MS spectrum) |
| Selectivity | Moderate | High (less interference) |
| Sensitivity | Very High | High, depending on resolution |
| Throughput | High | Moderate |
| Method Development | Requires transition tuning | Quick, minimal optimization |
| Data Reusability | No | Yes (retrospective) |
| Best Applications | High-throughput screening, routine quantification | Low-abundance targets, PTMs, validation |
Parallel Reaction Monitoring (PRM) has emerged as a complementary targeted technique performed on high-resolution, accurate-mass (HRAM) instruments like Orbitrap or Q-TOF systems [12]. While MRM monitors only predefined transitions on a triple quadrupole instrument, PRM captures the entire MS/MS spectrum of all resulting fragments using a high-resolution detector [12]. This allows for retrospective data analysis and increased confidence in analyte identification, making PRM especially useful for cases involving low-abundance targets, post-translational modifications, and samples with high background complexity [12]. However, MRM remains superior for high-throughput applications due to its faster cycle times and well-established standardized workflows [12].
Applications in Biomarker Validation
The Biomarker Development Pipeline
The biomarker development pipeline consists of several preclinical phases—discovery, verification, and validation—before final clinical evaluation [10] [13]. Biomarker discovery is typically performed using non-targeted "shotgun" proteomics approaches that provide relative quantitation of thousands of proteins in a small number of samples, yielding output as "up-or-down regulation" or "fold-increases" [10]. Following discovery, potential biomarker proteins undergo verification on sets of 10-50 patient samples to confirm their association with the disease state [10]. The final validation phase involves quantifying a small number of confirmed biomarkers across hundreds to thousands of samples to establish clinical utility [10] [13].
SRM/MRM plays a critical role in bridging the gap between biomarker discovery and validation [10] [13]. The technology's unique potential for reliable quantification of low-abundance analytes in complex mixtures makes it ideally suited for the verification phase [11]. Unlike discovery proteomics, which identifies potentially relevant proteins, SRM/MRM enables researchers to precisely measure predefined sets of candidate biomarkers across multiple samples with the consistency required for clinical application [11]. This targeted approach addresses a major limitation of conventional shotgun proteomics: the poor reproducibility of target selection, which often results in only partially overlapping sets of proteins being identified from similar samples [11].
Specific Applications in Clinical Proteomics
In clinical diagnostics and biomarker discovery, SRM/MRM techniques are increasingly adopted for the precise quantification of disease-associated biomarkers in biological fluids [7]. The accurate measurement of biomarker concentrations in clinical samples enables early disease detection, disease monitoring, and treatment optimization [7]. A key application is the absolute quantification of disease protein biomarkers in body fluids such as urine, which represents a critical step in the biomarker development pipeline [8]. The great advantage of targeted mass spectrometry-based methodologies like SRM/MRM is their capacity for accurate and specific simultaneous quantification of several biomarkers (multiplexing) using peptides as protein surrogates measured on triple quadrupole instruments [8].
For proteomics and protein quantification, SRM/MRM enables precise measurement of proteins across complex biological samples, providing insights into cellular processes, disease mechanisms, and therapeutic targets [7]. Researchers leverage these techniques to quantify protein expression levels, post-translational modifications, and protein-protein interactions with exceptional accuracy and sensitivity [7]. This application facilitates biomarker discovery, drug development, and personalized medicine by elucidating the dynamic proteome landscape [7]. Similarly, in metabolomics and small molecule analysis, the targeted quantification capabilities of SRM/MRM allow researchers to measure metabolite concentrations with high specificity and sensitivity, enabling comprehensive analysis of metabolic pathways, disease biomarkers, and drug metabolism [7].
Table: Performance Characteristics of SRM/MRM in Biomarker Validation
| Performance Measure | Typical Range | Context |
|---|---|---|
| Linear Range | 10^3 – 10^4 | For standard serum proteins [14] |
| Limit of Detection | 0.2 – 2 fmol | Comparable to commercial ELISA kits [14] |
| Reproducibility (R²) | 0.723 – 0.931 | Varies by protein/peptide [14] |
| Correlation with ELISA | R² = 0.565 – 0.928 | Depends on specific protein target [14] |
| Target Multiplexing | Up to 1000 transitions | Using scheduled SRM [11] |
Experimental Protocols for SRM/MRM Assay Development
Establishing a Targeted SRM/MRM Experiment
The development of a robust SRM/MRM assay involves a systematic approach that can be divided into distinct phases. The pre-mass spectrometry acquisition phase includes (1) generation of a target protein list, (2) selection of proteotypic peptides, and (3) experimental design [15]. The post-acquisition phase encompasses (1) peak detection, integration and quantification, (2) data quality assessment, (3) data visualization and exploratory analysis, and (4) fold change/statistical significance analysis [15].
The first critical step is the selection of a target protein set based on previous experiments, scientific literature, or various bioinformatics resources such as gene expression data, protein-protein interaction networks, or functionally related protein groups from gene ontology or KEGG databases [11]. For biomarker validation studies, this set typically derives from prior discovery experiments. In addition to the proteins of interest, several "housekeeping" proteins should be selected as an invariant reference set to correct for experimental variability such as uneven total protein amount per sample [11].
The next crucial step involves peptide selection for each targeted protein. Following tryptic digestion, proteins yield tens to hundreds of peptides, but typically only a few representative "proteotypic peptides" (PTPs) are targeted to infer the presence and quantity of a protein [11]. These peptides should exhibit good MS responses and uniquely identify the targeted protein or a specific isoform thereof [11]. Previous information from repositories like PeptideAtlas, Human Proteinpedia, GPM Proteomics Database, or PRIDE can help identify frequently observed peptides for proteins of interest [11]. Additional empirical rules for peptide selection include avoiding peptides with missed cleavages, methionine or cysteine residues (unless alkylated), and N-terminal glutamine or glutamate (which can form pyroglutamate), while favoring peptides between 7-20 amino acids in length [11].
Transition Optimization and Assay Validation
For each proteotypic peptide, the optimization of transitions is essential for developing a sensitive and specific SRM assay [11]. This process involves identifying fragment ions that provide optimal signal intensity and discriminate the targeted peptide from other species in the sample [11]. Transition development typically involves importing protein sequences into specialized software that generates preferred Q1 and Q3 m/z values based on parameters such as enzyme specificity, allowed missed cleavages, fixed modifications, and fragment types [14]. Filters are applied to narrow down the MRM transitions list, including Q1 m/z > 400, Q3 m/z < 1200, and peptide length between 6-30 amino acids [14].
The final transition list is refined by examining peak areas of candidate transitions after analyzing digested purified protein standards [14]. Typically, the top 3-4 peptides with the highest SRM/MRM response are selected as signature peptides, with 2-3 transitions monitored for each peptide to ensure specificity [14]. This approach creates easy-to-distinguish overlapping peaks with the same elution time in the chromatogram, confirming assay specificity [14].
Diagram: SRM/MRM Workflow on Triple Quadrupole Mass Spectrometer
Assay validation involves establishing key performance parameters including limit of detection (LOD), lower limit of quantification (LLOQ), linearity, carry-over, and specificity [15]. For "Response Curve" experiments, assays are typically performed in triplicates over multiple dilution ranges (e.g., seven points) to identify LLOQ, LOD, and linear range [15]. For "Mini-validation of Repeatability," experiments are performed in triplicates at three concentration levels ("High," "Medium," and "Low") repeated on five different days to approximate the variability of the assay in real-world practice [15]. Tools like MRMPlus can compute these analytical measures as recommended by the Clinical Proteomics Tumor Analysis Consortium (CPTAC) Assay Development Working Group for "Tier 2" assays—non-clinical assays sufficient to measure changes due to both biological and experimental perturbations [15].
Practical Implementation and Quality Control
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Essential Research Reagent Solutions for SRM/MRM Assay Development
| Reagent/Material | Function/Application | Examples/Specifications |
|---|---|---|
| Triple Quadrupole Mass Spectrometer | Targeted quantification of predefined transitions | Instrumentation with three quadrupole analyzers [7] |
| Liquid Chromatography System | Analyte separation prior to MS analysis | Nano-HPLC systems for improved sensitivity [14] |
| Trypsin (Sequencing Grade) | Protein digestion to generate peptides | Enzyme to protein ratio of 1:50 (w/w) [14] |
| Isotope-Labeled Peptide Standards | Absolute quantification internal standards | Heavy-labeled (D, 13C, 15N) peptides [9] [11] |
| Solid-Phase Extraction Columns | Sample cleanup and desalting | C18-based columns for peptide purification [16] |
| LC Separation Columns | Peptide separation prior to MS injection | ZORBAX 300SB-C18, 3.5μm, 150mm×0.075mm [14] |
| Quality Control Pools | Monitoring assay performance over time | Representative sample aliquots for longitudinal QC [10] |
Analytical Considerations and Quality Assurance
Successful implementation of SRM/MRM requires careful attention to several analytical factors. Instrument performance optimization involves parameters such as collision energy, cone voltage, and dwell times to maximize sensitivity and selectivity [7]. Chromatographic conditions, including mobile phase composition, flow rate, and column temperature, must be meticulously optimized to achieve efficient analyte separation and peak resolution [7]. Prior to sample analysis, instrument calibration and performance verification procedures are essential to ensure accurate quantification and data quality [7].
Scheduled SRM represents an important advancement for quantifying multiple proteins in a single experiment [11]. When targeting many proteins requiring numerous transitions, the dwell time for individual transitions is reduced, potentially compromising sensitivity [11]. Practical dwell-time settings range between 10 ms for good sensitivity and 100 ms for excellent sensitivity [11]. To ensure precise LC-MS quantification, at least eight data points should be acquired across the chromatographic elution profile of a peptide [11]. Assuming a peak width of 20 s at 10% peak height, a cycle time of 2 s is required, translating to approximately 200 transitions with 10 ms dwell time [11]. Scheduled SRM addresses this limitation by acquiring transitions for particular peptides only in time windows around their expected retention times, significantly increasing the number of peptides/proteins that can be detected and quantified in a single LC-MS experiment—potentially exceeding 1000 transitions with high sensitivity and reproducibility [11].
Diagram: Biomarker Pipeline with SRM/MRM Verification
Quality control and assessment are critical components of robust SRM/MRM assay development [15]. The complexities of biological matrices result in somewhat unpredictable analytical behavior of peptides and transitions, making experimental and analytical validation essential to establish that peptides and associated transitions serve as stable, quantifiable, and reproducible representatives of proteins of interest [15]. Open-source tools like MRMPlus streamline the assay development analytical workflow and minimize error predisposition by computing performance measures as recommended by the CPTAC Assay Development Working Group [15]. These tools accept inputs from preprocessing software like Skyline and generate performance-associated visualizations that provide global perspectives of assay performance across all assayed peptide transitions [15].
SRM and MRM mass spectrometry techniques represent powerful tools for targeted quantification in biomarker validation research. Their exceptional selectivity, sensitivity, and quantitative accuracy make them particularly valuable for verifying candidate biomarkers in complex biological matrices during the critical transition from discovery to clinical validation [7] [11]. The robust nature of these techniques, combined with their multiplexing capabilities, positions them as essential methodologies in the proteomics workflow, especially when traditional immunoaffinity-based techniques are limited by antibody availability [14].
As mass spectrometry technology continues to advance, SRM/MRM approaches remain foundational for precise protein quantification in systems biology and clinical applications [11]. The ability to reliably quantify predefined sets of proteins across multiple samples addresses a major limitation of discovery proteomics and supports the development of mathematical models that simulate biological systems and predict their behavior under different conditions [11]. Furthermore, the continuous development of standardized guidelines, open-source analytical tools, and quality control frameworks ensures that SRM/MRM assays will maintain their critical role in generating high-quality quantitative data for biomarker validation and drug development pipelines [15].
The journey of a biomarker from initial discovery to clinical validation is a complex, multi-stage process often described as a pipeline. This pathway is designed to systematically reduce a large number of candidate biomarkers identified through discovery efforts down to a select few that demonstrate robust clinical utility [17] [10]. The development of clinically useful biomarkers represents a critical bridge between basic research and patient care, enabling advancements in disease diagnosis, prognosis, therapeutic monitoring, and personalized medicine [18]. Despite the application of advanced "omics" technologies that generate hundreds to thousands of biomarker candidates, a discouragingly small number successfully navigate the entire pipeline to achieve regulatory approval and clinical implementation [17] [10]. This high attrition rate stems from numerous challenges, including the incredible mismatch between the volume of biomarker candidates and the scarcity of reliable assays and methods for proper validation [17]. The verification stage, in particular, represents a significant bottleneck—described as the "tar pit" of the pipeline—where many promising candidates fail due to inadequate analytical validation or insufficient clinical performance [17]. Understanding each phase of this pipeline, along with the appropriate technologies and methodologies required at each step, is essential for improving the success rate of biomarker development, particularly when utilizing powerful analytical platforms such as triple quadrupole mass spectrometry for targeted validation studies [4] [10].
Phases of the Biomarker Pipeline
Biomarker Discovery
The biomarker pipeline begins with the discovery phase, where potential biomarker candidates are identified through unbiased screening approaches [10] [18]. During this stage, researchers utilize various "omics" technologies—including genomics, proteomics, metabolomics, and lipidomics—to comprehensively profile biological samples from distinct clinical groups (e.g., diseased versus healthy controls) [18]. In proteomics-based discovery, mass spectrometry techniques are typically employed in a non-targeted "shotgun" approach to identify proteins that exhibit differential expression between comparative groups [10] [13]. These methods rely on relative quantitation, with results typically expressed as "up-or-down regulation" or "fold-increases" rather than absolute concentration measurements [10]. Common techniques include isobaric tagging (e.g., iTRAQ, TMT), label-free quantitation, and spectral counting, which enable the simultaneous comparison of thousands of proteins across multiple samples [10] [19]. The output of this phase is a extensive list of candidate biomarkers—often numbering in the hundreds or thousands—that show statistically significant associations with the disease or condition of interest [17] [10]. However, it is crucial to recognize that discovery efforts produce candidates (hypotheses), not validated biomarkers, and these efforts are inherently error-prone due to technological limitations, biological variability, and study design constraints [17].
Biomarker Prioritization and Verification
Following discovery, an essential prioritization step occurs to select the most promising candidates for further verification [17]. This stage involves filtering the extensive candidate list based on various biological and practical criteria, such as the magnitude of observed change, known biological relevance to the disease mechanism, likelihood of detection in accessible biofluids (e.g., plasma), and feasibility of developing robust assays for quantification [17] [10]. Proteins discovered in diseased tissues that are predicted to be secreted or located on the cell surface are often prioritized based on the assumption they might have greater access to circulation [17]. The subsequent verification phase represents a critical bottleneck in the biomarker pipeline, where prioritized candidates are assessed using targeted, quantitative methods in larger sample sets (typically 10-50 patients) [10] [20]. This stage aims to confirm the differential expression of candidates before committing resources to large-scale validation studies [17]. Targeted mass spectrometry approaches, particularly Multiple Reaction Monitoring (MRM) using triple quadrupole mass spectrometers, have emerged as powerful tools for biomarker verification due to their high sensitivity, specificity, and multiplexing capabilities [17] [4] [10]. Unlike discovery-phase approaches, verification requires absolute or precise relative quantitation to determine actual protein concentrations, moving beyond simple fold-change comparisons [10].
Clinical Validation and Qualification
The final preclinical phase involves rigorous clinical validation of verified biomarkers in well-designed, large-scale studies involving hundreds to thousands of patient samples [17] [10]. This stage assesses the clinical performance of the biomarker for its intended use, establishing diagnostic sensitivity and specificity, prognostic value, or predictive utility for treatment response [18]. The bar for clinical validation is exceptionally high, as biomarkers must demonstrate not only statistical significance but also clinical relevance and cost-effectiveness for their proposed application [17]. For example, a biomarker intended for population screening must meet extraordinarily high specificity standards to avoid excessive false positives, while a biomarker for diagnosing symptomatic patients may have different performance requirements [17]. The validation process must also evaluate whether the biomarker provides added value beyond existing standards of care [21]. Successful clinical validation leads to regulatory qualification, wherein biomarkers undergo review by regulatory agencies (e.g., FDA, EMA) against stringent criteria for analytical validity, clinical validity, and clinical utility [22] [18]. This process requires extensive documentation, including assay validation data, clinical trial evidence, and proof of clinical significance [18].
Table 1: Key Stages in the Biomarker Development Pipeline
| Pipeline Stage | Primary Objective | Sample Size | Key Technologies | Output |
|---|---|---|---|---|
| Discovery | Identify candidate biomarkers | Small (n < 50) | Shotgun proteomics, LC-MS/MS, Isobaric tagging | List of 100s-1000s of candidate proteins with fold changes |
| Prioritization | Filter candidates based on biological/practical criteria | N/A | Literature mining, bioinformatics, preliminary testing | Prioritized list of 10s of candidates |
| Verification | Confirm differential expression | Moderate (n = 10-50) | Targeted MS (MRM), immunoassays | Verified shortlist of candidates with quantitative data |
| Validation | Assess clinical performance | Large (n = 100s-1000s) | Validated assays (MS or immunoassays), clinical trials | Clinically validated biomarker with performance characteristics |
| Qualification | Regulatory approval | Very large (n = 1000s+) | GLP-compliant assays, multi-center trials | Approved biomarker for specific clinical context |
Mass Spectrometry in Biomarker Verification and Validation
Triple Quadrupole Mass Spectrometry and Multiple Reaction Monitoring (MRM)
Triple quadrupole (QqQ) mass spectrometers have become the cornerstone technology for biomarker verification and validation due to their exceptional sensitivity, specificity, and quantitative capabilities [4] [10]. These instruments operate through a tandem mass spectrometry approach where the first quadrupole (Q1) selects specific precursor ions, the second quadrupole (Q2) fragments these ions through collision-induced dissociation, and the third quadrupole (Q3) filters for specific product ions [4]. This configuration enables Multiple Reaction Monitoring (MRM)—a highly specific targeted analysis method that monitors predetermined precursor-product ion transitions corresponding to peptides of interest [10]. The exceptional selectivity of MRM allows for precise quantification of target analytes even in complex biological matrices like plasma or serum, making it ideally suited for biomarker verification studies [17] [10]. The strength of MRM lies in its ability to multiplex, simultaneously monitoring dozens to hundreds of candidate biomarkers in a single analysis, thus significantly accelerating the verification process [17]. Furthermore, the incorporation of stable isotope-labeled standard (SIS) peptides enables absolute quantification, providing precise concentration measurements essential for clinical assay development [10]. The robust nature, relatively low cost, and wide dynamic range of triple quadrupole instruments have established them as the preferred platform for targeted biomarker quantification in both research and clinical settings [4].
Advanced MS Techniques: TMT-MRM and Isobaric Tagging
Recent technological advancements have combined the multiplexing advantages of isobaric tagging with the sensitivity and specificity of MRM, creating powerful integrated approaches for biomarker verification [19]. Isobaric tagging methods, such as Tandem Mass Tags (TMT) and Isobaric Tags for Relative and Absolute Quantitation (iTRAQ), enable simultaneous analysis of multiple samples (2-11 plex) by labeling them with chemical tags that have identical overall mass but yield distinct reporter ions upon fragmentation [19]. When coupled with MRM, these techniques significantly enhance throughput and quantitative precision while reducing analytical variation [19]. For example, TMT labeling applied to phospholipid analysis allowed comprehensive screening of 196 human plasma samples from Alzheimer's disease cohorts with only 40 MRM measurements, dramatically improving efficiency without compromising data quality [19]. This integrated approach is particularly valuable for large-scale verification studies where analyzing hundreds of samples for dozens of candidates would be prohibitively time-consuming and expensive using conventional methods [19]. The method demonstrates high reproducibility in human plasma and enables direct comparison of biomarker levels across multiple patient cohorts, facilitating robust biomarker candidate assessment [19].
Table 2: Comparison of Mass Spectrometry Approaches in Biomarker Development
| Parameter | Discovery (Shotgun Proteomics) | Verification (MRM/QqQ) | Validation (Targeted MS) |
|---|---|---|---|
| Primary Goal | Comprehensive protein identification | Targeted candidate verification | Precise quantification for clinical use |
| Quantitation Type | Relative (fold-changes) | Absolute or precise relative | Absolute concentration |
| Multiplexing Capacity | 1000s of proteins | 10s-100s of targets | Typically < 10 targets |
| Sample Throughput | Low to moderate | High with multiplexing | High for targeted assays |
| Key Strengths | Unbiased, comprehensive | Specific, sensitive, quantitative | Robust, reproducible, validated |
| Limitations | Semi-quantitative, limited dynamic range | Requires a priori knowledge | Limited scope, extensive validation required |
Experimental Protocols
Protocol: MRM-Based Biomarker Verification Using Triple Quadrupole MS
Objective: To verify candidate protein biomarkers in plasma/serum samples using targeted MRM mass spectrometry.
Materials and Reagents:
- Triple quadrupole mass spectrometer with nanoflow or conventional LC system
- Stable Isotope-Labeled Standard (SIS) peptides for each target protein
- Trypsin (sequencing grade) for protein digestion
- Solid-phase extraction cartridges (C18) for sample cleanup
- Mobile phase solvents: water and acetonitrile with 0.1% formic acid
Sample Preparation Procedure:
- Protein Extraction and Digestion:
- Deplete high-abundance proteins from plasma/serum using immunoaffinity columns [23].
- Reduce proteins with 5 mM dithiothreitol (56°C, 30 min) and alkylate with 15 mM iodoacetamide (room temperature, 30 min in dark).
- Digest proteins with trypsin (1:20-1:50 enzyme-to-protein ratio) at 37°C for 12-16 hours.
- Add known quantities of SIS peptides before digestion (if quantifying pre-digestion analytes) or after digestion (if quantifying peptides).
- Peptide Cleanup:
- Desalt digested peptides using C18 solid-phase extraction cartridges.
- Dry samples under vacuum and reconstitute in 0.1% formic acid for LC-MS analysis.
LC-MRM/MS Analysis:
- Chromatographic Separation:
- Use reversed-phase C18 column (e.g., 15-25 cm length, 75 μm inner diameter).
- Apply linear gradient from 2% to 35% acetonitrile over 30-60 minutes at 200-300 nL/min flow rate.
MRM Method Development:
- Select 2-3 proteotypic peptides per protein (typically 7-20 amino acids long).
- For each peptide, identify 3-5 optimal fragment ions for monitoring.
- Optimize collision energies for each transition.
- Schedule MRM transitions within specific retention time windows to maximize monitoring capacity.
Data Acquisition:
- Set dwell times to achieve 8-12 data points across each chromatographic peak.
- Use unit resolution in both Q1 and Q3.
- Include heavy isotope-labeled internal standards for each target peptide.
Data Analysis:
- Peak Integration and Review:
- Integrate chromatographic peaks for all transitions using Skyline or similar software.
- Apply quality controls: co-elution of light and heavy peptides, consistent retention times, and matching relative intensities of fragment ions.
- Quantification:
- Calculate peak area ratios of light (natural) to heavy (SIS) peptides.
- Generate calibration curves using stable isotope-labeled standards for absolute quantification.
- Determine protein concentrations based on peptide standards.
This protocol enables specific, sensitive, and reproducible quantification of candidate protein biomarkers, providing crucial data for decision-making before proceeding to large-scale validation studies [17] [10] [20].
Protocol: TMT-MRM Multiplexed Biomarker Verification
Objective: To simultaneously verify multiple candidate biomarkers across several sample groups using TMT labeling combined with MRM.
Materials and Reagents:
- Tandem Mass Tag (TMT) 6-plex or 11-plex reagents
- Aminoxy TMT reagents for lipid analyses [19]
- Triethylamine solution (10% in THF)
- Ethanolamine solution (10% in THF) for quenching
- Acetic acid (10% in THF) for neutralization
Procedure:
- Sample Preparation and Labeling:
- Extract lipids or digest proteins from patient samples as described in Section 4.1.
- Dissolve extracted samples in 50 μL tetrahydrofuran (THF).
- Add 9 μL of TMT reagent (pre-dissolved in acetonitrile) to each sample.
- Add 5 μL of triethylamine solution (10% in THF) to catalyze the reaction.
- Incubate at room temperature for 20 hours.
Reaction Quenching and Pooling:
- Add 5 μL of ethanolamine solution (10% in THF) to quench the reaction.
- Incubate for 10 minutes.
- Add 5 μL of acetic acid (10% in THF) to neutralize samples.
- Combine TMT-labeled samples in desired multiplexing scheme (e.g., 6-plex).
MRM Analysis:
- Develop MRM methods targeting TMT-labeled analytes of interest.
- Monitor specific transitions that include TMT reporter ions (e.g., m/z 126-131 for 6-plex).
- Use pooled quality control samples labeled with a distinct TMT channel for normalization.
This multiplexed approach significantly enhances throughput while maintaining quantitative accuracy, making it particularly valuable for large-scale verification studies [19].
Workflow Visualization
Biomarker Pipeline with MS Approaches
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagent Solutions for Biomarker MS Workflows
| Reagent/Material | Function | Application Examples |
|---|---|---|
| TMT/iTRAQ Reagents | Isobaric mass tags for multiplexed relative quantification | Simultaneous analysis of 2-11 samples; biomarker verification studies [19] |
| Stable Isotope-Labeled Standards (SIS) | Internal standards for absolute quantification | Precise measurement of protein/peptide concentrations in MRM assays [10] |
| Immunoaffinity Depletion Columns | Remove high-abundance proteins | Reduce dynamic range in plasma/serum samples to enhance detection of low-abundance biomarkers [23] |
| Trypsin (Sequencing Grade) | Proteolytic enzyme for protein digestion | Generates peptides for bottom-up proteomics; essential for MRM assay development [10] |
| Phospholipid Removal Plates | Extract lipids from biological samples | Sample preparation for lipidomic biomarker discovery and verification [19] |
| C18 Solid-Phase Extraction Plates | Desalt and concentrate peptide samples | Sample cleanup before LC-MS analysis; improves signal-to-noise ratio [20] |
TMT-MRM Experimental Workflow
Quality Assurance and Control in Biomarker Validation
Ensuring the reliability and reproducibility of biomarker assays requires rigorous quality assurance and control measures throughout the validation process [20]. Fit-for-purpose (FFP) biomarker assay validation has emerged as a guiding principle, where validation requirements are aligned with the proposed context of use (COU) for any given biomarker [22]. This approach recognizes that the level of validation needed for early research phases differs substantially from that required for clinical decision-making or regulatory submission [22]. Key analytical performance characteristics that must be established include precision, accuracy, sensitivity, specificity, and reproducibility [22] [20]. For mass spectrometry-based biomarker assays, specific challenges include the presence of endogenous analytes in control matrices and difficulties in procuring appropriate reference standards that accurately represent the endogenous molecules [22]. Implementing quality control samples at multiple levels—including process controls, internal standards, and pooled quality control samples—is essential for monitoring assay performance and ensuring data reliability [20] [19]. Additionally, proper study design elements such as sample blinding, randomization, and batch effect control are critical for minimizing bias and ensuring the validity of study conclusions [21] [20]. As biomarkers progress toward clinical implementation, adherence to regulatory guidelines and standards becomes increasingly important, with detailed documentation of assay validation data and clinical evidence required for regulatory qualification [22] [18].
The biomarker development pipeline represents a rigorous, multi-stage process designed to translate promising research findings into clinically useful tools for precision medicine. Despite the challenges and high attrition rates, systematic approaches that leverage appropriate technologies at each stage—particularly triple quadrupole mass spectrometry for verification—can significantly improve the efficiency and success of biomarker development [17] [4]. The integration of advanced methodologies such as TMT-MRM further enhances throughput and quantitative precision, helping to address the critical bottleneck in biomarker verification [19]. As mass spectrometry technologies continue to evolve and standardization improves, these platforms are poised to play an increasingly important role in bridging the gap between biomarker discovery and clinical validation, ultimately accelerating the delivery of novel biomarkers to improve patient care [17] [4] [23].
Why QqQ is the Gold Standard for Targeted Quantification
The triple quadrupole mass spectrometer (QqQ) maintains its position as the gold standard for targeted quantitative analysis in biomedical research due to its exceptional sensitivity, specificity, and robustness. Within the critical context of biomarker validation, QqQ systems, particularly when operating in Selected Reaction Monitoring (SRM) or Multiple Reaction Monitoring (MRM) modes, provide the precise and reproducible data required to translate potential biomarkers from discovery into clinically applicable tools [24] [4]. This application note details the operational principles, experimental protocols, and specific applications that solidify the QqQ's status for targeted quantification in biomarker research and drug development.
The triple quadrupole mass spectrometer, first developed in the late 1970s by Enke and Yost, consists of three sequentially arranged quadrupole mass analyzers [1] [25]. The first (Q1) and third (Q3) quadrupoles act as mass filters, capable of selecting ions based on their mass-to-charge ratio (m/z). The second quadrupole (q2) is a radio-frequency (RF)–only collision cell that fragments the precursor ions selected by Q1 through collision-induced dissociation (CID) with an inert gas [1]. This linear arrangement of components, often abbreviated QqQ, enables a tandem-in-space configuration that is ideal for targeted analyses [1].
The Pillars of QqQ Quantification
The supremacy of QqQ in targeted quantification rests on three fundamental pillars, which are particularly crucial for the biomarker validation pipeline where reliability is paramount.
Unmatched Sensitivity and Specificity
The core strength of the QqQ lies in its two-stage mass filtering process. Q1 selects a specific precursor ion, excluding the vast majority of chemical background. After fragmentation in q2, Q3 monitors a specific product ion. This double mass selection creates a highly specific ion transition, dramatically reducing background noise and leading to superior signal-to-noise ratios [24] [25]. This configuration increases sensitivity by one to two orders of magnitude compared to full-scan methods, enabling the detection of low-abundance biomarkers in complex matrices like plasma or urine [24].
Robust and Reliable Quantification
QqQ systems provide a wide linear dynamic range and excellent analytical precision [26]. The predictable and efficient fragmentation in the RF-only collision cell, coupled with the unit mass resolution of the quadrupole mass filters, yields highly reproducible results essential for absolute quantitation [10]. This robustness makes QqQ the preferred platform for high-throughput clinical applications, including newborn screening programs where it is used to detect metabolic biomarkers for congenital diseases [4].
Operational and Economic Efficiency
Despite the emergence of high-resolution mass spectrometers (HRMS), QqQ systems remain more affordable and cost-effective for dedicated quantitative analyses [27] [4]. They are relatively easy to operate and maintain, making them accessible to a broad range of clinical and research laboratories. The number of biomedical studies utilizing QqQ has increased 2–3 times this decade, demonstrating its growing adoption and utility [4].
Table 1: QqQ Performance in Key Application Areas
| Application Area | Key Metric | Performance/Role |
|---|---|---|
| Newborn Screening [4] | Utilization Rate in Studies | 84% (924 out of 1098 studies) |
| Endocrine Testing [4] | Trend | Increasing adoption as reference method, displacing immunoassays |
| Biomarker Validation [24] [10] | Primary Mode | Selected Reaction Monitoring (SRM) / Multiple Reaction Monitoring (MRM) |
| General Quantitative Analysis [26] | Benefits | Increased selectivity, improved S/N, lower limits of quantitation, wider linear range |
The Biomarker Validation Pipeline and QqQ
The journey of a protein biomarker from discovery to clinical use is a multi-stage process with distinct analytical requirements at each phase. QqQ mass spectrometry plays an indispensable role in the verification and validation stages.
The Biomarker Pipeline
The pipeline begins with discovery, typically using non-targeted "shotgun" proteomics on high-resolution instruments to identify hundreds of potential protein candidates from a small number of samples [10]. This is followed by verification, where QqQ-based SRM assays are deployed to screen tens to hundreds of these candidate proteins across a larger set of samples (e.g., 10-50 patients) [10]. The final preclinical stage is validation, where a small number of the most promising biomarkers are quantified across hundreds of samples using highly optimized SRM assays on QqQ platforms [10]. The final clinical validation involves analyzing these biomarkers across 500–1000+ samples [10].
Diagram 1: The Biomarker Validation Pipeline. QqQ-MS is critical for the verification and validation phases.
QqQ Scan Modes for Biomarker Analysis
The flexibility of the QqQ instrument is demonstrated through its various scan modes, each serving a distinct purpose in quantitative and qualitative analysis [1].
Table 2: Key Operational Modes of a QqQ Mass Spectrometer
| Scan Mode | Q1 Function | Q3 Function | Primary Application in Biomarker Research |
|---|---|---|---|
| Selected/Multiple Reaction Monitoring (SRM/MRM) [1] | Selects specific precursor ion | Selects specific product ion | Targeted quantification of known biomarkers with high sensitivity and specificity. |
| Product Ion Scan [1] | Selects specific precursor ion | Scans all product ions | Obtaining fragmentation patterns for structural elucidation and transition selection. |
| Precursor Ion Scan [1] [28] | Scans all precursor ions | Selects specific product ion | Selective detection of all precursors that fragment to yield a common product ion (e.g., a characteristic functional group). |
| Neutral Loss Scan [1] [28] | Scans all precursor ions | Scans with a constant mass offset | Detection of all precursors that undergo a common neutral loss (e.g., H₂O, NH₃). |
Diagram 2: QqQ Operational Modes. Different scanning configurations support various analytical needs.
Detailed Protocol: Biomarker Verification via SRM on a QqQ Platform
The following protocol outlines a standardized workflow for verifying candidate protein biomarkers in human plasma using LC-SRM on a QqQ mass spectrometer.
Experimental Workflow
Diagram 3: SRM Experimental Workflow. Key steps for targeted biomarker verification.
Step-by-Step Methodology
Step 1: Sample Preparation
- Protein Extraction: Extract proteins from human plasma or serum samples. Utilize techniques like Solid-Phase Extraction (SPE) for removing abundant proteins and lipids to reduce sample complexity [27].
- Protein Digestion: Digest the protein extract into peptides using a sequence-specific protease, most commonly trypsin. Use appropriate buffers (e.g., ammonium bicarbonate) and control digestion conditions (time, temperature, enzyme-to-substrate ratio) to ensure complete and reproducible digestion [24] [10].
Step 2: Selection of Proteotypic Peptides and Transitions
- Proteotypic Peptide (PTP) Selection: For each candidate biomarker protein, select 1-3 peptides that uniquely identify the protein (proteotypic). These peptides should exhibit strong ionization efficiency and not contain unstable or modifiable amino acids [24]. Computational tools and databases like PeptideAtlas can aid in this selection [24].
- Transition Selection: For each PTP, select 2-4 optimal fragment ions to monitor. Singly charged y-type ions are typically the most abundant and stable fragments generated by CID in a QqQ [24]. Select transitions where the fragment ion has a larger m/z than the precursor ion to minimize chemical background [24].
Step 3: Liquid Chromatography (LC) Separation
- Separate the digested peptide mixture using reversed-phase liquid chromatography (e.g., C18 column) or hydrophilic interaction liquid chromatography (HILIC) [27].
- Use a binary gradient with mobile phase A (e.g., water/0.1% formic acid) and B (e.g., acetonitrile/0.1% formic acid) to achieve optimal peptide separation and co-elution of the target peptide with its stable isotope-labeled internal standard.
Step 4: SRM/MRM Assay on QqQ
- Instrument Parameters:
- Ion Source: Electrospray Ionization (ESI) is most common.
- Q1/Q3 Resolution: Set to unit resolution (0.7 Da FWHM).
- Dwell Time: 10-50 ms per transition, adjusted to ensure sufficient data points across the chromatographic peak.
- Collision Energy: Optimized for each transition; can be predicted from precursor charge and m/z [27].
- Data Acquisition: Monitor the predefined precursor-product ion transitions for each PTP across their expected retention time windows.
Step 5: Data Analysis and Quantification
- Integrate the chromatographic peaks for each transition.
- Use a stable isotope-labeled internal standard (SIL) for each target peptide for absolute quantification. The SIL peptide is identical in sequence but contains heavy isotopes (e.g., 13C, 15N), ensuring identical chemical behavior [10].
- Calculate the ratio of the peak area of the native peptide to the peak area of the SIL peptide. Use a calibration curve, prepared by spiking known amounts of the SIL peptide into the sample matrix, to determine the absolute concentration of the target protein [10].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Essential Research Reagents and Materials for QqQ-based Biomarker Assays
| Reagent/Material | Function/Application | Example Specifications |
|---|---|---|
| Trypsin, Sequencing Grade [10] | Proteolytic enzyme for digesting proteins into peptides for bottom-up proteomics. | High purity to minimize autolysis; modified to reduce self-digestion. |
| Stable Isotope-Labeled (SIL) Peptides [10] | Internal standards for absolute quantification; correct for sample prep and ionization variability. | Synthesized with >97% purity and heavy amino acids (e.g., [13C6, 15N2]-Lysine). |
| LC-MS Grade Solvents [27] | Mobile phases for liquid chromatography; high purity is critical to reduce background noise. | Acetonitrile, Methanol, Water with low volatile impurities. |
| Abundant Protein Depletion Kit [10] | Immunoaffinity columns to remove high-abundance proteins (e.g., albumin) from plasma/serum. | Spin columns or cartridges designed for specific sample volumes. |
| Solid-Phase Extraction (SPE) Plates [27] | For sample clean-up and removal of phospholipids and other interferents post-digestion. | 96-well format for high-throughput; chemistries like C18 or EMR-Lipid. |
Application Example: Protocol for Analysis of New Psychoactive Substances
This protocol, adapted from Rodrigues de Morais et al. (2020), demonstrates the use of QqQ scan modes for qualitative screening and quantitative analysis of NBOMe and NBOH compounds in blotter papers [28].
Sample Preparation:
- Extraction: Cut a segment of the blotter paper and extract with 1 mL of methanol in an ultrasonic bath for 15 minutes.
- Preparation: Dilute the extract 1:100 with mobile phase for LC-MS/MS analysis.
LC-MS/MS Analysis:
- Chromatography: Use a C18 column with a gradient of water and acetonitrile, both with 0.1% formic acid.
- Initial Screening (Precursor Ion Scan):
- Set Q3 to monitor a characteristic product ion (e.g., m/z 121 for NBOMes).
- Scan Q1 over a mass range (e.g., m/z 300-500) to identify all precursors that generate this fragment.
- Confirmation (Product Ion Scan):
- For each potential precursor found, set Q1 to its m/z.
- Scan Q3 to obtain a full product ion spectrum for library matching and structural confirmation.
- Quantification (SRM/MRM):
- For confirmed analytes, develop a quantitative SRM method.
- Use two transitions per compound for confident identification (primary for quantification, secondary for qualification).
- Use deuterated internal standards if available.
Key Instrument Parameters:
- Ion Source: ESI, positive mode.
- Gas Temperature: 300°C
- Nebulizer Gas: 7 L/min
- Collision Gas: Nitrogen or Argon
This multi-mode approach showcases the power of QqQ for comprehensive analysis, from unknown screening to precise quantification.
The triple quadrupole mass spectrometer remains the undisputed gold standard for targeted quantification in biomarker validation and drug development. Its unparalleled sensitivity, derived from the dual mass filtering of SRM/MRM, combined with its robustness, quantitative linear dynamic range, and operational efficiency, makes it an indispensable tool for generating high-quality, reproducible data. As biomarker research continues to drive advances in precision medicine, the QqQ platform will remain a cornerstone technology for verifying and validating the next generation of diagnostic and prognostic biomarkers.
The adoption of triple quadrupole (QqQ) mass spectrometry in biomedical research represents a paradigm shift towards highly specific, quantitative analysis in biomarker discovery and validation. This technology, particularly through multiple reaction monitoring (MRM) and selected reaction monitoring (SRM), has become the cornerstone of modern proteomic pipelines seeking to translate candidate biomarkers into clinically applicable diagnostic tools [10]. The inherent complexity of biological systems and the critical need for precision in drug development have accelerated the integration of QqQ mass spectrometry, moving beyond qualitative identification to robust, reproducible quantification [20]. This transition is vital, as the biomarker development pipeline demands progressively higher levels of analytical stringency—from initial discovery in small cohorts to final clinical validation in populations of hundreds or thousands [10]. The rising adoption of QqQ methodologies is fundamentally linked to their ability to bridge this gap, offering the specificity, sensitivity, and multiplexing capacity required to verify and validate protein biomarkers with the rigor demanded by regulatory agencies [20] [29]. This protocol outlines the application of QqQ mass spectrometry within this context, providing a detailed framework for its use in targeted proteomic studies aimed at biomarker validation.
Current Landscape and Quantitative Data
The use of mass spectrometry in biomedical research, especially for protein biomarker analysis, has seen substantial growth and formal recognition. Key quantitative data and trends are summarized in the tables below.
Table 1: Adoption Metrics for Analytical Techniques in Biomarker Research
| Metric | Value / Trend | Context & Notes |
|---|---|---|
| Primary MS Technique for Verification | Multiple Reaction Monitoring (MRM) | Bridges gap between discovery and validation; considered the gold standard for verification [10]. |
| Number of FDA-Approved Protein Biomarkers (as of 2014) | ~24 for cancer | Highlights the challenge of translation; many are based on immunoassays, a target for MS-based replacement [10]. |
| Dynamic Range of Newer Immunoassays (e.g., MSD) | Up to 5 orders of magnitude | Shows performance of alternative techniques; QqQ MS offers comparable or superior specificity [29]. |
| Sample Throughput in Discovery vs. Validation | Discovery: 10s of samplesVerification: 10-50 samplesValidation: 100-500+ samples | Illustrates the inverse relationship between sample number and proteins quantified; QqQ MRM is optimized for the high-sample-number phases [10]. |
Table 2: Key Performance Characteristics of QqQ MRM versus Immunoassays
| Characteristic | QqQ MRM/MS | Immunoassay (e.g., ELISA) |
|---|---|---|
| Specificity | High (based on precursor ion mass + fragment ion mass) | High (dependent on antibody affinity and specificity) [29]. |
| Multiplexing Capability | High (can monitor 100s of proteins in a single run) | Low to Moderate (multiplexing requires multiple, compatible antibodies) [29]. |
| Assay Development Time | Relatively short (weeks) | Long (months to years for antibody production and validation) [29]. |
| Critical Reagents | Synthetic stable isotope-labeled peptides | Target protein standard and matched antibody pairs [29]. |
| Susceptibility to Matrix Effects | Can be mitigated with internal standards | Can be affected by cross-reactivity with homologous or endogenous proteins [29]. |
Experimental Protocol: QqQ MRM for Biomarker Verification
This protocol details the steps for verifying a panel of candidate protein biomarkers in human plasma or serum using liquid chromatography-coupled QqQ mass spectrometry (LC-MRM/MS).
Sample Preparation
Objective: To reproducibly process complex biofluids (e.g., plasma, serum) to generate peptides for LC-MRM/MS analysis.
Materials:
- Plasma/Serum Samples: From well-characterized cohorts (e.g., case-control). Aliquot and store at -80°C.
- Digestion Buffer: 50 mM Tris-HCl, pH 8.0-8.5.
- Reducing Agent: 10 mM Dithiothreitol (DTT).
- Alkylating Agent: 25 mM Iodoacetamide (IAA).
- Protease: Sequencing-grade modified trypsin.
- Solid-Phase Extraction (SPE) Plates: e.g., C18 resin for desalting and peptide cleanup.
- Internal Standards: Heavy isotope-labeled synthetic peptides (e.g., ( ^{13}C/^{15}N)-labeled) for each target protein (AQUA peptides).
Procedure:
- High-Abundance Protein Depletion: Deplete major proteins (e.g., albumin, IgG) from plasma/serum using immunoaffinity columns per manufacturer's instructions. This step is optional but highly recommended for deeper analysis.
- Protein Denaturation, Reduction, and Alkylation:
- Dilute 10-20 µL of plasma/serum (or depleted equivalent) with 100 µL of digestion buffer.
- Add DTT to a final concentration of 10 mM. Incubate at 60°C for 30 minutes.
- Cool to room temperature. Add IAA to a final concentration of 25 mM. Incubate in the dark for 30 minutes.
- Proteolytic Digestion:
- Add trypsin at a 1:20 to 1:50 (w/w) enzyme-to-protein ratio.
- Incubate at 37°C for 12-16 hours.
- Quench the reaction by acidifying with 1% formic acid (FA).
- Peptide Cleanup:
- Desalt the digested peptide mixture using C18 SPE plates. Elute peptides with 50% acetonitrile (ACN)/0.1% FA.
- Dry the eluents completely in a vacuum concentrator.
- Spike-in of Internal Standards:
- Reconstitute the dried peptide pellet in an appropriate volume of 0.1% FA containing a known concentration of the heavy isotope-labeled peptide internal standards.
- Centrifuge at high speed to remove any insoluble material before LC-MS analysis.
LC-MRM/MS Method Development and Data Acquisition
Objective: To establish a highly specific and sensitive MRM assay for target peptides and acquire quantitative data.
Materials:
- Nano-flow or High-flow LC System: Configured with a C18 reverse-phase column.
- QqQ Mass Spectrometer: e.g., Agilent 6495, Sciex 7500, or Thermo Scientific TSQ Altis.
- Mobile Phase A: 0.1% Formic acid in water.
- Mobile Phase B: 0.1% Formic acid in acetonitrile.
Procedure:
- Peptide Selection: From your candidate protein list, select 2-3 proteotypic peptides per protein that are unique, 7-20 amino acids long, and avoid post-translational modification sites or ragged ends.
- Transition Selection:
- For each target peptide, use Skyline or similar software to predict precursor ion (m/z) and 3-5 most intense fragment ions (y- or b-series).
- Synthesize the light and heavy peptides to empirically confirm and optimize transitions.
- Define the final MRM method, including retention time, precursor ion > fragment ion transitions, and optimal collision energies for each transition.
- Liquid Chromatography:
- Inject a fixed volume of the prepared sample.
- Separate peptides using a binary gradient, e.g., from 3% to 35% Mobile Phase B over 30-60 minutes.
- Mass Spectrometry Data Acquisition:
- Operate the QqQ mass spectrometer in MRM mode.
- Set Q1 and Q3 to unit resolution.
- Schedule MRMs based on the known retention time of each peptide to maximize the number of data points acquired across each peak.
Data Analysis and Quantification
Objective: To extract and analyze MRM data to determine the relative or absolute concentration of target proteins.
Materials:
- Software: Skyline, MRM Peaktyper, or MultiQuant.
Procedure:
- Peak Integration: Import raw data into analysis software. Manually review and integrate peaks for all light (endogenous) and heavy (internal standard) transitions.
- Quality Control:
- Confirm the co-elution of light and heavy peptide peaks.
- Ensure the relative intensities of fragment ions for the light peptide match those of the heavy internal standard (within ~20%).
- Quantitative Calculation:
- For absolute quantification, calculate the ratio of the peak area of the light peptide to the peak area of the heavy internal standard for each peptide.
- Use a calibration curve (from serial dilutions of heavy peptide) to convert this ratio into a molar concentration.
- For relative quantification (e.g., diseased vs. control), calculate the light-to-heavy ratio and perform statistical comparison between sample groups.
Workflow Visualization
Biomarker Pipeline with QqQ
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for QqQ MRM Biomarker Studies
| Reagent / Material | Function / Role | Critical Notes |
|---|---|---|
| Stable Isotope-Labeled Standard (SIS) Peptides | Internal standards for precise quantification; corrects for sample prep losses and MS variability. | Essential for absolute quantification. Must be identical in sequence and behavior to the target endogenous peptide [10]. |
| Sequencing-Grade Modified Trypsin | Proteolytic enzyme for digesting proteins into measurable peptides. | High purity and specificity are critical for reproducible and complete digestion [20]. |
| Immunoaffinity Depletion Columns | Remove high-abundance proteins from plasma/serum to enhance detection of lower-abundance biomarkers. | Increases dynamic range and reduces signal suppression [20]. |
| LC Columns (C18, nano-flow) | Separate complex peptide mixtures prior to MS analysis. | Column performance directly impacts sensitivity and resolution [20]. |
| Quality Control (QC) Sample | Monitors instrument performance and reproducibility across the entire batch of samples. | Typically a pooled sample from all study aliquots or a commercial standard [20]. |
From Theory to Practice: Developing Robust QqQ Assays for Biomarker Verification
In the pipeline of biomarker validation, targeted mass spectrometry, particularly using triple quadrupole instruments, has emerged as the method of choice for verifying and quantifying candidate proteins with high specificity and sensitivity [30]. The core of this targeted approach lies in the precise selection of proteotypic peptides (PTPs)—peptides that uniquely represent a target protein or protein isoform and can be consistently detected by mass spectrometry [31] [32]. Subsequently, the development of optimal ion transitions for Selected Reaction Monitoring (SRM) or Multiple Reaction Monitoring (MRM) is crucial for achieving the high sensitivity and quantitative accuracy required to detect low-abundance biomarkers in complex matrices [33] [30]. This protocol details a systematic workflow for building a robust targeted assay, from in silico peptide selection to experimental optimization of MS parameters, specifically framed within the context of biomarker validation research.
The Role of Proteotypic Peptides in Biomarker Validation
Proteotypic peptides are the cornerstone of targeted proteomics for biomarker verification. Their fundamental property is to act as a surrogate for the parent protein, enabling its unambiguous identification and quantification [31]. In the context of a complex biological sample, such as plasma or urine, a well-chosen PTP must be unique to the protein of interest, thereby avoiding false positives from other proteins in the background proteome [34]. The use of PTPs also confers significant practical advantages. Searching MS/MS spectra against a database containing only proteotypic peptides, rather than the entire proteome, can reduce data analysis time by 20-fold due to the drastic reduction in database size [32]. Furthermore, concentrating on this most-observable subset of peptides can implicitly reduce the likelihood of false identifications [35].
Workflow for Developing a Targeted SRM/MRM Assay
The process of developing a targeted assay is multi-staged, involving both computational and experimental components. The following diagram illustrates the complete workflow from initial candidate selection to a finalized, quantitative assay.
Figure 1: A complete workflow for developing a targeted SRM/MRM assay, from protein candidate list to a validated quantitative method.
Selecting Proteotypic Peptides (PTPs)
Core Criteria for Selection
The initial selection of peptides is a computational process aimed at identifying the most suitable peptides to represent the target protein.
- Uniqueness to the Protein: The peptide sequence must be unique to the target protein or a specific isoform within the background proteome of the sample organism (e.g., human) to ensure the assay's specificity [31] [34].
- Consistent Observability: PTPs should be peptides that are consistently observed in mass spectrometry experiments, indicating favorable ionization and detection properties [35] [32].
- Ideal Physicochemical Properties: Peptides should typically be between 7 and 25 amino acids long. Shorter peptides may lack specificity, while longer ones may exhibit poor ionization and fragmentation [34].
- Avoidance of Problematic Sequences:
- Methionine and Cysteine: These residues are prone to oxidation, which can lead to multiple forms and complicate quantification [34].
- N-terminal Glutamine: This can cyclize to form pyro-glutamic acid, leading to non-quantitative conversion [34].
- Consecutive Prolines: Can cause peak broadening in chromatography, reducing sensitivity [34].
- Missed Tryptic Cleavages: Peptides with predictable and complete tryptic cleavage are preferred for reproducible generation [34].
Practical Selection Methodology
A practical approach to PTP selection can be derived from the mining of unique proteomes, such as that of Pyrococcus furiosus, for ideal peptide tags. The following table summarizes key selection criteria used in such a study [34].
Table 1: Criteria for Selecting Optimal Proteotypic Peptides for Quantification (PQS Peptides)
| Selection Criterion | Rationale | Action |
|---|---|---|
| Unique to Background Proteome | Ensures assay specificity for the target protein. | Map peptide against all known model organism protein databases. |
| High Ion Coverage | Indicates efficient fragmentation and multiple detectable fragments. | Select peptides with an average ion coverage of 30 or higher. |
| High Differential Score | Reflects strong LC-MS signal intensity and observability. | Filter for peptides with a differential score >6 (or >2 with 3+ spectral counts). |
| No M, C, N-term Q | Avoids oxidation and non-quantitative pyro-glu formation. | Discard peptides containing these residues. |
| No Missed Cleavages | Ensures reproducible and complete digestion. | Filter for perfect tryptic peptides. |
| No Consecutive Prolines | Prevents chromatographic peak broadening. | Discard peptides with Pro-Pro motifs. |
Transition Selection and MS Parameter Optimization
Selecting Optimal Fragment Ions
Once PTPs are selected, the next step is to determine the best precursor-to-fragment ion transitions for MRM monitoring.
- Ion Type Preference: In triple quadrupole systems using Collision-Induced Dissociation (CID), singly charged y-type ions are the most predominant and stable fragments. b-type ions and doubly charged fragments are significantly less stable [30].
- Fragment Selection: Choose 2-4 fragment ions per peptide to enable confident identification and quantification. Ions should be greater than 3 amino acids in length [33].
- Specificity and Signal-to-Noise: Select product ions with an m/z greater than the precursor ion's m/z. This helps avoid chemical background noise, which typically produces fragments with smaller m/z, thereby improving the signal-to-noise ratio [30] [36]. Avoid common neutral losses (e.g., water, ammonia) as primary transitions, as they are less unique [36].
Optimizing Instrument Parameters
Generalized equations for parameters like collision energy (CE) provide a starting point, but individual optimization is often necessary for maximum sensitivity [33].
Table 2: Key MS Parameters for Optimization in Targeted Assays
| Parameter | Function | Optimization Goal |
|---|---|---|
| Collision Energy (CE) | Controls energy for CID fragmentation; too much can destroy the target ion. | Find the voltage that generates the most abundant signal for the desired product ion [33] [36]. |
| Cone Voltage (CV) / Declustering Potential (DP) | Removes solvent and gas molecules clustered around the ion before the first mass filter. | Optimize for a clear precursor ion signal and maximal transmission of the desired ion [33]. |
| Ion Source Parameters (e.g., Gas Temp., Nozzle Voltage) | Governs the efficiency of droplet desolvation and analyte ionization. | Use statistical design (e.g., Plackett-Burman, Box-Behnken) to find optimal values for maximum response [37]. |
A highly efficient strategy for optimizing CE and CV is to test multiple parameter values in a single LC-MS run. This is accomplished by creating a method with subtly adjusted precursor and product m/z values (e.g., at the hundredth decimal place) for the same transition, each coded to trigger a different CE or CV value. This workflow avoids run-to-run variability and allows for rapid determination of the optimal setting for each transition [33]. The diagram below details this process.
Figure 2: A workflow for the rapid optimization of collision energy (CE) for an MRM transition in a single run, avoiding run-to-run variability.
Table 3: Key Research Reagent Solutions for Targeted Assay Development
| Reagent / Resource | Function in Assay Development |
|---|---|
| Stable Isotope Labeled (SIL) Peptides | Synthesized with heavy isotopes (e.g., 13C, 15N); act as internal standards for precise quantification, correcting for ionization suppression and sample preparation losses [34]. |
| Immobilized Trypsin | Provides efficient and complete protein digestion without enzyme contamination, which is crucial for accurate quantification of cross-linked peptides like ε-(γ-glutamyl) lysine [38]. |
| Proteotypic Peptide Libraries | Curated databases (e.g., PeptideAtlas, GPM) of peptides known to be consistently observed for specific proteins, providing a starting point for PTP selection [35] [32]. |
| Universal Peptide Tags (PQS) | Peptides selected from unique proteomes (e.g., P. furiosus) that are absent in model organisms; can be engineered into proteins as quantitative tags for absolute quantification in co-IP studies [34]. |
Concluding Protocol: A Practical Example
This section outlines a concrete protocol for developing an MRM assay for a hypothetical biomarker candidate, "Protein X," in human plasma.
In Silico Peptide Selection:
- Input the protein sequence of "Protein X" into a database like PeptideAtlas or use a tool like SpectraST to generate a list of potential proteotypic peptides [33].
- Apply the filters listed in Table 1 (length, uniqueness, absence of problematic residues) to select 3-5 candidate peptides.
Transition Selection:
- For each candidate peptide, use spectral libraries or in silico prediction tools to identify the 4-5 most intense singly charged y-ions.
- Ensure the chosen product ions have m/z values higher than the precursor m/z.
Synthesis and Preliminary Testing:
- Synthesize the candidate peptides (light and heavy labeled versions).
- Infuse each peptide to confirm its MS/MS spectrum and select the top 3-4 most intense and unique fragments for the final transition list.
Parameter Optimization:
- Use the single-run optimization workflow (Figure 2) to determine the optimal CE and CV for each of the final transitions.
- For ion source parameters, employ an experimental design (e.g., Box-Behnken) to systematically optimize factors like nozzle voltage, sheath gas flow, and temperature for maximum overall sensitivity [37].
Assay Validation:
- Spike the heavy labeled peptides into a series of plasma samples.
- Establish a calibration curve by analyzing samples with known concentrations of the analyte.
- Validate the assay for sensitivity (LOD, LOQ), precision (<20% CV is acceptable [38]), linearity (r > 0.99 [37]), and specificity (no interfering peaks in the matrix).
By rigorously following this detailed protocol, researchers can build targeted mass spectrometry assays that are specific, sensitive, and robust, thereby providing reliable data for the critical step of biomarker validation.
Advanced Sample Preparation for Complex Matrices like Blood Plasma
In the context of biomarker validation for triple quadrupole mass spectrometry research, the preparation of complex matrices such as blood plasma is a pivotal pre-analytical step. The accuracy, sensitivity, and reproducibility of mass spectrometric analysis are profoundly influenced by the quality of the sample preparation process [39]. Blood plasma, while a rich source of potential protein and peptide biomarkers, presents significant analytical challenges due to its immense complexity and extreme dynamic range of protein concentrations, which can exceed ten orders of magnitude [40] [39]. A handful of highly abundant proteins, such as serum albumin and immunoglobulins, constitute nearly 90% of the total protein content by weight, effectively masking the detection of lower-abundance proteins that may serve as critical disease biomarkers [40]. Without robust and standardized preparation and fractionation protocols, important biological information can be lost in the background noise, compromising the downstream biomarker validation pipeline [40] [20]. This document outlines detailed application notes and protocols designed to address these challenges, providing researchers and drug development professionals with methodologies to ensure reliable and reproducible plasma sample preparation for rigorous biomarker validation studies using triple quadrupole mass spectrometry.
Blood Collection: Choosing Between Plasma and Serum
The decision to use plasma or serum is fundamental and must align with the research objectives. Although they are often used interchangeably in biochemical tests, plasma and serum yield different protein profiles, and insufficient information exists to definitively recommend one over the other for all proteomic studies [40].
- Plasma is the liquid component of blood, obtained by collecting blood into tubes containing an anticoagulant. The blood does not clot, and cells are removed via centrifugation [41]. The choice of anticoagulant is critical, as it can influence the sample's stability and interfere with subsequent analysis.
- Serum resembles plasma in composition but lacks coagulation factors. It is obtained by allowing a blood specimen to clot prior to centrifugation [41] [40].
Table 1: Comparison of Blood Collection Tube Types
| Tube Color | Anticoagulant/Additive | Sample Type | Key Considerations |
|---|---|---|---|
| Lavender | EDTA | Plasma | Slightly less stable over long periods; chelating action may help prevent coagulation [41] [40]. |
| Blue | Citrate | Plasma | Liquid form dilutes plasma; platelets are more stable [41] [40]. |
| Green | Heparin | Plasma | Relatively stable; can bind to numerous proteins and may be contaminated with endotoxin, potentially stimulating cytokine release [41] [40]. |
| Grey/Yellow | Potassium Oxalate/Sodium Fluoride | Plasma | [41] |
| Red | None (clot activator) | Serum | Standard for serum collection [41]. |
| Red with black | Gel separator | Serum | Gel facilitates separation of clot from serum [41] [40]. |
Core Protocol: Plasma Preparation from Whole Blood
The following protocol is adapted from standardized procedures for processing whole blood into plasma [41].
Materials and Reagents
- Anticoagulant-treated blood collection tubes (e.g., Lavender-top EDTA, Light Blue-top citrate, or Green-top heparin) [41].
- Refrigerated centrifuge.
- Pasteur pipettes or micropipettes.
- Clean polypropylene tubes for plasma storage.
- Safety equipment: gloves, lab coat, eye protection.
Step-by-Step Procedure
- Collection: Collect whole blood via venipuncture directly into a commercially available anticoagulant-treated tube. Gently invert the tube 8-10 times immediately after collection to ensure proper mixing with the anticoagulant [41].
- Centrifugation: Place the tube in a refrigerated centrifuge (2-8°C) and spin at 1,000–2,000 x g for 10 minutes to separate cells from the plasma. For applications requiring platelet-poor plasma, centrifuge at 2,000 x g for 15 minutes [41].
- Plasma Extraction: Using a Pasteur pipette, carefully extract the supernatant (plasma), ensuring not to disturb the cell pellet. Immediately transfer the plasma into a clean, labeled polypropylene tube. Maintain samples at 2–8°C throughout this handling process [41].
- Aliquoting and Storage: If the plasma is not analyzed immediately, aliquot it into 0.5 mL portions in cryovials to avoid repeated freeze-thaw cycles. Store aliquots at –20°C for short-term use or –80°C for long-term preservation [41].
Note: Hemolyzed, icteric, or lipemic samples can invalidate certain tests and should be noted [41].
Advanced Processing for Biomarker Research
The immense dynamic range of the plasma proteome necessitates advanced processing to detect low-abundance biomarkers. The core workflow involves depletion, digestion, and cleanup prior to mass spectrometry.
Depletion of High-Abundance Proteins
Removing highly abundant proteins like albumin and immunoglobulins is crucial to reduce dynamic range and unmask lower-abundance species [39].
- Principle: Immunoaffinity techniques use antibodies immobilized on resins to selectively bind and remove specific high-abundance proteins from the plasma sample [39].
- Protocol Considerations:
- Commercial immunoaffinity depletion kits are widely available.
- A significant drawback is that abundant proteins often bind to other proteins, which could result in the co-depletion of protein complexes containing low-abundance proteins of interest [39].
Protein Denaturation, Reduction, and Alkylation
This step prepares proteins for enzymatic digestion into peptides, which are more suitable for LC-MS/MS analysis [39].
- Denaturation: Proteins are denatured using strong chaotropic agents like urea or thiourea to unfold their structure [39].
- Reduction: Disulfide bonds are irreversibly broken using reducing agents such as dithiothreitol (DTT) or Tris(2-carboxyethyl)phosphine (TCEP) [39].
- Alkylation: The free sulfhydryl groups on cysteine residues are then alkylated with reagents such as iodoacetamide to prevent reformation of disulfide bonds [39].
Enzymatic Digestion
Digestion fragments proteins into peptides, which ionize and fragment more efficiently in the mass spectrometer [39].
- Principle: An endoproteinase, most commonly trypsin, is used to hydrolytically break peptide bonds at specific amino acid sequences [39].
- Protocol (In-Solution Digestion): The denatured, reduced, and alkylated protein mixture is incubated with trypsin at an optimized ratio (e.g., 1:50 enzyme-to-protein) and temperature (typically 37°C) for several hours to overnight to achieve complete digestion [39].
Peptide Cleanup and Desalting
Samples must be cleaned to remove salts, detergents, and other interferents that can suppress ionization and impair MS analysis [39].
- Principle: Solid-phase extraction (SPE) using C18 cartridges or tips is the standard method. Peptides bind to the hydrophobic resin, while salts and other polar contaminants are washed away. Peptides are then eluted with an organic solvent like acetonitrile [39].
- Protocol: Follow manufacturer instructions for the specific SPE product. The eluted peptides are then dried down in a vacuum concentrator and reconstituted in a mass spectrometry-compatible solvent (e.g., 0.1% formic acid) for LC-MS/MS analysis.
The following workflow diagram synthesizes the core and advanced protocols into a single, coherent process from blood draw to MS-ready sample:
Diagram 1: Workflow for plasma sample preparation for MS.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Plasma Proteomics
| Item Category | Specific Examples | Function | Considerations for Biomarker Studies |
|---|---|---|---|
| Anticoagulants | EDTA, Citrate, Heparin [41] | Prevents blood clotting for plasma preparation. | EDTA chelates metals; Heparin can bind proteins and contain endotoxin [41] [40]. |
| Protease Inhibitors | Commercial cocktails (e.g., AEBSF, Leupeptin) | Protects extracted proteins from degradation by endogenous proteases. | Critical for preserving the native proteome state; add to lysis buffer [39]. |
| Depletion Kits | Immunoaffinity columns for Albumin/IgG removal | Reduces sample complexity by removing highly abundant proteins. | Risk of co-depleting bound low-abundance proteins; standardizes dynamic range [39]. |
| Chaotropes | Urea, Thiourea | Denatures proteins, making them more accessible to enzymatic digestion. | Must be removed prior to digestion to avoid enzyme inactivation [39]. |
| Reducing Agents | DTT, DTE, TCEP | Breaks disulfide bonds within and between proteins. | TCEP is more stable and effective than DTT [39]. |
| Alkylating Agents | Iodoacetamide | Irreversibly blocks cysteine thiol groups post-reduction. | Prevents reformation of disulfides; must be used in the dark [39]. |
| Proteases | Trypsin, Lys-C | Digests proteins into peptides for LC-MS/MS analysis. | Trypsin is the gold standard; enzyme-to-substrate ratio and incubation time are key [39]. |
| SPE Cartridges | C18, C8 | Desalts and concentrates peptide samples prior to MS. | Removes ion-suppressing salts and buffers; essential for robust LC-MS performance [39]. |
Standardization of key parameters is essential for inter-laboratory reproducibility. The following table summarizes critical quantitative data for plasma preparation and handling.
Table 3: Key Parameters for Plasma Sample Handling
| Process Parameter | Recommended Conditions | Notes & Variations |
|---|---|---|
| Centrifugation Speed | 1,000 - 2,000 x g [41] | Standard force for plasma separation from cells. |
| Centrifugation Time | 10 minutes [41] | Sufficient for clear separation. |
| Centrifugation Temperature | 2 - 8°C (refrigerated) [41] | Maintains sample stability. |
| Post-Centrifugation Handling | 2 - 8°C [41] | Maintain samples on ice or in a chilled rack. |
| Aliquot Volume | 0.5 mL [41] | Minimizes freeze-thaw cycles. |
| Short-term Storage | -20°C or lower [41] | |
| Long-term Storage | -80°C [41] | Preserves sample integrity for extended periods. |
| Platelet Depletion Spin | 2,000 x g for 15 minutes [41] | For applications requiring platelet-poor plasma. |
Case Study: LC-MS/MS Method for a Specific Biomarker
A study developing a liquid chromatography-triple quadrupole mass spectrometry (LC-MS/MS) method for the determination of isopeptide ε-(γ-glutamyl) lysine in human urine provides an excellent example of an optimized sample preparation workflow for a specific biomarker, with principles directly applicable to plasma [42].
- Biomarker Significance: ε-(γ-glutamyl) lysine is a cross-link formed by transglutaminase 2 (TG2) activity, which is upregulated in fibroproliferative diseases like pulmonary fibrosis and chronic kidney disease. Quantifying this isopeptide in biofluids has significant potential as a diagnostic or monitoring biomarker [42].
- Sample Preparation Challenge: Traditional methods used a complex multi-enzyme digestion in solution, requiring high enzyme levels that contaminated the analysis and lacked the sensitivity and selectivity needed for low-abundance targets in a complex matrix [42].
- Innovative Solution: The researchers systematically addressed these challenges by implementing:
- Optimized Protein Precipitation: For efficient concentration and buffer exchange.
- Immobilized Enzyme Digestion Kit: Utilizing a series of endo- and exo-peptidases covalently anchored on silica nanoparticles. This enabled complete digestion without enzyme contamination, as the enzymes could be removed by centrifugation [42].
- LC-MS/MS Detection with MRM: The use of a triple quadrupole mass spectrometer in Multiple Reaction Monitoring (MRM) mode provided the necessary selectivity and sensitivity, achieving a limit of detection of 0.1 ng/mL in human urine with a precision of less than 20% CV [42].
This case highlights how tailored sample preparation, moving from traditional solution-based enzymes to modern immobilized enzyme technology, is critical for the successful validation of challenging biomarkers using triple quadrupole MS.
In the context of biomarker validation research using triple quadrupole mass spectrometry, matrix effects represent a critical analytical challenge that can compromise data integrity. Matrix effects are defined as the suppression or enhancement of a target analyte's ionization efficiency caused by co-eluting compounds present in the biological sample matrix [43] [44]. These effects are particularly problematic in quantitative bioanalysis for biomarker validation, as they can lead to inaccurate measurements, reduced sensitivity, and poor reproducibility [45]. In liquid chromatography-mass spectrometry (LC-MS), matrix effects primarily occur when endogenous matrix components—such as phospholipids, salts, metabolites, and proteins—co-elute with the target analyte and interfere with its ionization in the mass spectrometer source [44] [45]. The electrospray ionization (ESI) technique, widely used in triple quadrupole MS for biomarker research, is especially susceptible to these effects compared to atmospheric pressure chemical ionization (APCI) due to differences in ionization mechanisms [43] [45]. Understanding and mitigating matrix effects is therefore essential for generating reliable, accurate, and reproducible biomarker data that meets rigorous validation standards.
Understanding Matrix Effect Mechanisms in LC-MS/MS
The mechanisms underlying matrix effects differ significantly between ionization techniques. In ESI, which operates via ionization in the liquid phase followed by transfer of charged species to the gas phase, matrix effects primarily occur through several pathways [44] [45]. Matrix components can compete for available charge in the electrospray droplets, reducing the ionization efficiency of target analytes. They can also alter droplet properties by increasing viscosity and surface tension, which impedes solvent evaporation and gas-phase ion release. Additionally, co-eluting compounds may coprecipitate with analytes or neutralize gas-phase ions [45].
In contrast, APCI is generally less susceptible to matrix effects because ionization occurs primarily in the gas phase rather than in solution [43]. The table below compares the characteristics of these ionization techniques regarding matrix effect susceptibility.
Table 1: Comparison of Ionization Techniques and Matrix Effect Susceptibility
| Ionization Technique | Ionization Mechanism | Susceptibility to Matrix Effects | Primary Sources of Interference |
|---|---|---|---|
| Electrospray Ionization (ESI) | Charge transfer in liquid phase, followed by droplet evaporation and gas-phase ion release | High | Phospholipids, salts, ion-pairing agents, metabolites with similar polarity |
| Atmospheric Pressure Chemical Ionization (APCI) | Evaporation to gas phase followed by chemical ionization using corona discharge | Moderate | Compounds with high proton affinity (positive mode) or gas-phase acidity (negative mode) |
The extent of matrix effects is compound-specific and depends on the chemical properties of both the analyte and interfering matrix components [45]. Phospholipids are particularly problematic in ESI-based analysis of plasma and serum samples, often causing significant ion suppression for a wide range of analytes [44]. The composition of biological matrices varies considerably, meaning matrix effects must be evaluated for each specific sample type and analytical method [45].
Experimental Protocols for Matrix Effect Assessment
Post-Column Infusion Method for Qualitative Assessment
The post-column infusion method provides a qualitative assessment of matrix effects across the chromatographic run, identifying regions of ion suppression or enhancement [43] [46].
Materials and Reagents:
- LC-MS/MS system with triple quadrupole mass spectrometer
- T-piece connector for post-column infusion
- Syringe pump for constant analyte delivery
- Blank biological matrix (e.g., drug-free plasma, urine)
- Standard solution of target analyte in appropriate solvent
- Mobile phase components (HPLC grade)
Procedure:
- Connect the T-piece between the LC column outlet and the MS interface.
- Set up the syringe pump to deliver a constant flow (typically 5-20 µL/min) of analyte standard solution through the T-piece.
- Inject a processed blank matrix sample onto the LC column while maintaining the post-column infusion.
- Perform chromatographic separation using the intended method conditions.
- Monitor the analyte signal throughout the chromatographic run.
Interpretation: A constant signal indicates no matrix effects. Signal depression at specific retention times indicates regions of ion suppression, while signal increases indicate ion enhancement [43] [46]. This method provides a "matrix effect chromatogram" that highlights problematic retention time windows requiring chromatographic optimization.
Post-Extraction Spike Method for Quantitative Assessment
The post-extraction spike method provides a quantitative measurement of matrix effects for specific analytes [43].
Materials and Reagents:
- Blank matrix from at least six different sources
- Standard solutions of target analytes at low, medium, and high concentrations
- Stable isotope-labeled internal standards (when available)
- All solvents and reagents for sample preparation
Procedure:
- Prepare two sets of samples:
- Set A: Spike analyte standards into neat solution (mobile phase or solvent)
- Set B: Spike the same concentrations of analyte standards into extracted blank matrix
- Process both sets according to the standard sample preparation protocol.
- Analyze all samples by LC-MS/MS using the validated method.
- Calculate the matrix effect (ME) for each analyte using the formula: ME (%) = (Peak Area Set B / Peak Area Set A) × 100
Interpretation: ME values <100% indicate ion suppression, >100% indicate ion enhancement, and ≈100% indicate no significant matrix effects. The FDA Bioanalytical Method Validation guidelines recommend evaluating matrix effects using blank matrix from at least six different sources to account for biological variability [45].
Table 2: Matrix Effect Assessment Methods and Applications
| Assessment Method | Type of Information | Advantages | Limitations |
|---|---|---|---|
| Post-Column Infusion | Qualitative identification of suppression/enhancement regions | Identifies problematic retention times; No blank matrix required | Does not provide quantitative data; Labor-intensive for multiple analytes |
| Post-Extraction Spike | Quantitative measurement for specific analytes | Provides numerical matrix effect values; Uses actual sample preparation method | Requires blank matrix; Single concentration level assessment |
| Slope Ratio Analysis | Semi-quantitative screening across concentration ranges | Evaluates matrix effects at multiple concentration levels | Requires blank matrix; More complex implementation |
Strategic Approaches to Minimize Matrix Effects
Chromatographic Optimization Strategies
Mobile Phase and Gradient Optimization: Adjusting chromatographic conditions represents one of the most effective approaches for mitigating matrix effects. The goal is to achieve temporal separation of analytes from major matrix interferences [43] [46]. This can be accomplished by:
- Extending run times to widen the separation window
- Modifying gradient profiles to shift analyte retention times away from problematic regions identified by post-column infusion
- Adjusting mobile phase pH to alter analyte retention, particularly for ionizable compounds
- Using alternative buffer systems (e.g., ammonium acetate/formate instead of phosphate buffers) that are more compatible with MS detection
Column Selection: Column chemistry significantly impacts selectivity and matrix component separation [46]. Consider:
- Alternative stationary phases (e.g., HILIC, phenyl-hexyl, pentafluorophenyl) that provide different selectivity compared to conventional C18 columns
- Longer column dimensions (e.g., 150 mm vs. 50 mm) to enhance resolution
- Smaller particle sizes (e.g., sub-2μm) to improve efficiency without increasing analysis time
Sample Preparation Techniques
Selective sample preparation effectively removes matrix components before chromatographic separation [43] [44] [45].
Protein Precipitation: While simple and rapid, protein precipitation provides minimal cleanup of phospholipids and other endogenous compounds, often resulting in significant matrix effects [45].
Liquid-Liquid Extraction (LLE): LLE can effectively remove phospholipids and other non-polar interferents, particularly when using selective organic solvents matched to analyte polarity [45].
Solid-Phase Extraction (SPE): SPE offers superior cleanup capabilities through multiple mechanisms:
- Reverse-phase SPE effectively removes non-polar interferents
- Ion-exchange SPE targets ionic matrix components
- Mixed-mode SPE combines multiple retention mechanisms for enhanced selectivity
- Selective sorbents including molecularly imprinted polymers (MIPs) provide highly specific extraction, though commercial availability may be limited [43]
Internal Standardization Approaches
The use of appropriate internal standards represents a critical strategy for compensating for residual matrix effects [43] [44].
Stable Isotope-Labeled Internal Standards (SIL-IS): These are the gold standard for compensating matrix effects because they:
- Exhibit nearly identical chemical properties and chromatography as the native analyte
- Experience the same degree of ion suppression/enhancement as the analyte
- Co-elute with the native analyte, ensuring matched matrix effects
- Allow for accurate quantification even when matrix effects cannot be eliminated [44]
Structural Analog Internal Standards: When SIL-IS are unavailable, structurally similar compounds can be used, though they may not perfectly match analyte behavior in the presence of matrix effects [43].
The following diagram illustrates the strategic decision-making process for addressing matrix effects in method development:
Research Reagent Solutions for Matrix Effect Management
Table 3: Essential Research Reagents and Materials for Matrix Effect Investigation
| Reagent/Material | Function in Matrix Effect Studies | Application Notes |
|---|---|---|
| Blank Biological Matrix | Baseline for post-extraction spike experiments; preparation of matrix-matched standards | Should be sourced from at least 6 different lots to account for variability [45] |
| Stable Isotope-Labeled Internal Standards | Compensation for matrix effects during quantification; method validation | Ideally deuterated or 13C-labeled analogs that co-elute with native analytes [44] |
| SPE Cartridges (Various Chemistries) | Selective removal of matrix interferences prior to analysis | C18 for non-polar analytes; mixed-mode for ionic compounds; HLB for broad-spectrum cleanup |
| LC Columns (Alternative Phases) | Enhanced chromatographic separation of analytes from matrix components | HILIC for polar compounds; phenyl-hexyl for aromatics; pentafluorophenyl for specific selectivity |
| Post-column Infusion T-piece | Connection for continuous analyte infusion during matrix effect assessment | Must be low-dead-volume and compatible with LC flow rates |
| Mobile Phase Additives (MS-compatible) | Modification of chromatographic selectivity without compromising ionization | Ammonium acetate/formate instead of non-volatile salts; acidic modifiers for positive mode |
Application to Biomarker Validation in Triple Quadrupole MS
In biomarker validation research using triple quadrupole mass spectrometry, controlling matrix effects is particularly crucial due to the quantitative nature of these studies and the regulatory standards they must meet [45]. The following protocol outlines a comprehensive approach to address matrix effects in biomarker assay development and validation.
Comprehensive Matrix Effect Assessment Protocol for Biomarker Validation
Experimental Design:
- Source Selection: Procure blank matrix from at least six different sources to account for biological variability [45].
- Concentration Levels: Prepare quality control samples at low, medium, and high concentrations covering the validated range.
- Sample Processing: Process samples using the proposed extraction method alongside neat standards in solvent.
Assessment Criteria:
- Matrix Factor (MF): Calculate using the formula MF = Peak response in matrix / Peak response in solvent
- Precision: The coefficient of variation (CV%) of the matrix factor should be <15% across different matrix lots
- Internal Standard Normalization: Evaluate whether stable isotope-labeled internal standards effectively compensate for variability
Documentation: Record matrix effects for each analyte and each matrix lot, identifying any trends related to specific matrix components or sample processing techniques.
Integrated Workflow for Matrix Effect Control
The following diagram illustrates a comprehensive workflow for managing matrix effects throughout the biomarker validation process:
Effective management of matrix effects is fundamental to developing robust LC-MS/MS methods for biomarker validation using triple quadrupole mass spectrometry. A systematic approach combining appropriate sample preparation, optimized chromatographic separation, and effective internal standardization provides the most reliable strategy for controlling these effects. The protocols and strategies outlined in this application note enable researchers to identify, quantify, and mitigate matrix effects, thereby ensuring the generation of accurate and reproducible biomarker data that meets rigorous validation standards. As biomarker research continues to advance toward analyzing increasingly complex biological samples at lower concentrations, diligent attention to matrix effects remains essential for maintaining data quality and scientific integrity.
The validation of protein biomarkers is a critical step in translating basic research into clinically useful diagnostics. Within this landscape, triple quadrupole mass spectrometry (QqQ MS) has emerged as a cornerstone technology due to its superior sensitivity, specificity, and quantitative capabilities. Operating primarily in Multiple Reaction Monitoring (MRM) mode, also referred to as Selected Reaction Monitoring (SRM), QqQ MS enables highly precise absolute quantification of target analytes in complex biological matrices [10] [47]. This technique isolates predefined precursor ions in the first quadrupole, fragments them in the second, and monitors specific product ions in the third, creating a highly selective "ion transition" that minimizes background interference [48] [47]. The typical biomarker development pipeline proceeds through several preclinical phases—discovery, verification, and validation—before final clinical evaluation [10]. While discovery often uses non-targeted "shotgun" methods to analyze thousands of proteins in a small number of samples, verification and validation employ targeted MS approaches like MRM to precisely quantify a smaller panel of candidate biomarkers across hundreds of samples [10] [20]. This targeted approach is indispensable for establishing clinically relevant protein concentrations, making QqQ MS an invaluable tool for developing robust biomarker panels in oncology, nephrology, and endocrinology [4] [3] [47].
Application Note 1: Cancer Biomarker Panels
Context and Rationale
Cancer remains a leading cause of mortality worldwide, creating an urgent need for biomarkers for early detection, disease classification, prediction of therapeutic response, and treatment monitoring [10] [47]. The complexity of cancer biology necessitates the use of biomarker panels rather than reliance on single proteins [10]. While numerous potential cancer biomarkers have been discovered, very few have been translated into clinically approved tests, highlighting the critical importance of robust verification and validation workflows [10] [47]. Liquid biopsies, which analyze cancer-derived signals in biofluids like blood and urine, offer a less invasive source for these biomarkers and enable longitudinal monitoring [49] [47].
Exemplary Study and Data
Targeted proteomics using QqQ MS has emerged as a powerful platform for quantifying cancer protein biomarkers with advantages over traditional immunoassays, including the ability to multiplex and avoid antibody cross-reactivity issues [47]. A prominent example of a successful multi-protein panel is the OVA1 test, which combines five protein biomarkers to assess the risk of ovarian malignancy [10] [50]. The following table summarizes key characteristics of FDA-approved protein biomarker panels and related tests for cancer.
Table 1: Clinically Implemented Protein Biomarker Panels and Tests for Cancer
| Test/Biomarker Panel | Clinical Use | Cancer Type | Specimen | Methodology |
|---|---|---|---|---|
| OVA1 (Multiple proteins) | Prediction of malignancy [10] | Ovarian [10] | Serum [10] | Immunoassay [10] |
| ROMA (HE4 + CA-125) | Prediction of malignancy [10] | Ovarian [10] | Serum [10] | Immunoassay [10] |
| Circulating Tumor Cells (EpCAM, CD45, cytokeratins 8, 18+, 19+) | Prediction of progression/survival [10] | Breast [10] | Whole Blood [10] | Immunomagnetic capture/immunofluorescence [10] |
| PromarkerD (CD5L, APOA4, IBP3) | Prediction of diabetic kidney disease [50] | (Non-cancer application shown for methodology comparison) | Plasma [50] | Immunoaffinity MRM-MS [50] |
Detailed Protocol: MRM-Based Quantification of Cancer Biomarkers from Plasma
Principle: This protocol describes the absolute quantification of a panel of candidate protein biomarkers from human plasma or serum using liquid chromatography-tandem mass spectrometry (LC-MRM/MS) with stable isotope-labeled (SIS) peptides as internal standards [47].
Workflow Diagram:
Step-by-Step Procedure:
Sample Preparation:
- Thaw plasma/serum samples on ice and centrifuge at 14,000 × g for 10 minutes to remove precipitates [47].
- Immunodepletion: To overcome the vast dynamic range of plasma proteins, use an immunoaffinity column (e.g., Human 14 Multiple Affinity Removal System, MARS14) to remove the top 14 most abundant proteins according to the manufacturer's protocol [48] [47]. This step is crucial for detecting lower-abundance cancer biomarkers.
Protein Digestion:
- Measure the protein concentration of the immunodepleted flow-through.
- Add a known amount of SIS protein or "winged" SIS peptide standard to the sample before digestion. This corrects for variability introduced during the digestion process [47].
- Denature, reduce disulfide bonds (e.g., with TCEP), and alkylate cysteine residues (e.g., with iodoacetamide) [50].
- Digest the proteins into peptides using sequencing-grade trypsin (typically at a 1:50 enzyme-to-protein ratio) overnight at 37°C [48].
Peptide Clean-up:
LC-MRM/MS Analysis:
- Separate the peptides using reversed-phase nano-liquid chromatography (nanoLC) with a C18 column and a gradient of increasing organic solvent (acetonitrile) [48] [47].
- Analyze the eluting peptides using a triple quadrupole mass spectrometer.
- The MS is programmed to monitor specific transitions (precursor ion > product ion) for both the endogenous "light" peptide and its co-eluting SIS "heavy" peptide for each biomarker. At least two transitions per peptide are monitored: one as the "quantifier" and another as the "qualifier" to confirm identity [47].
Data Analysis and Quantification:
- Process the raw data using targeted proteomics software (e.g., Skyline).
- The peak area ratio of the endogenous peptide to the SIS peptide is calculated for the quantifier transition.
- Absolute quantification is achieved by interpolating this ratio against a calibration curve prepared with known concentrations of the authentic standard [47].
Application Note 2: Diabetic Kidney Disease (DKD) Biomarker Panels
Context and Rationale
Diabetic kidney disease is a major complication of diabetes, with one in three adult diabetics affected [48] [50]. The current gold standard diagnostics—urinary albumin-to-creatinine ratio (ACR) and estimated glomerular filtration rate (eGFR)—have limitations in reliability and predictive power [48] [51]. This creates a significant need for more robust and specific biomarkers for early detection of DKD, enabling intervention before irreversible kidney damage occurs [48] [51].
Exemplary Study and Data
A comprehensive MS-based workflow has been successfully applied to discover and validate a plasma biomarker panel for DKD. The study involved a discovery phase using isobaric tagging for relative and absolute quantitation (iTRAQ) on pooled plasma samples, followed by verification and validation using MRM on a large cohort of 572 patients [48]. This process yielded a validated panel of five proteins significantly associated with DKD. In a separate study, the PromarkerD test was developed, which combines the measurement of three plasma proteins (CD5L, APOA4, IBP3) with clinical variables (age, HDL-cholesterol, eGFR) in a predictive algorithm [50]. The transition of PromarkerD from a research-grade immunodepletion-MRM assay to a high-throughput clinical-grade immunoaffinity-MRM assay demonstrates the potential for translating QqQ MS-based biomarker panels into clinical practice [50].
Table 2: Mass Spectrometry-Based Biomarker Panels for Diabetic Kidney Disease
| Biomarker Panel / Signature | Biological Source | Key Components | Performance / Clinical Utility |
|---|---|---|---|
| DKD Panel (iTRAQ/MRM) [48] | Plasma | 5-protein panel | Significantly associated with albuminuria, eGFR, and CKD stage (ROC AUC = 0.77) [48] |
| PromarkerD [50] | Plasma | CD5L, APOA4, IBP3 (plus clinical variables) | Predicts development of DKD 4 years in advance [50] |
| Urinary Peptidomic Classifier [51] | Urine | 273 peptides (CKD273) / Collagen fragments, SERPINA1, SERPING1 | Predicts progression from normo- to macroalbuminuria; identifies progressors to reduced eGFR independent of albuminuria [51] |
Detailed Protocol: Immunoaffinity-MRM Assay for a Multiplexed Protein Panel
Principle: This protocol describes a higher-throughput, robust immunoaffinity capture method coupled to MRM for quantifying a specific protein panel (e.g., PromarkerD). It leverages antibody specificity for enrichment and MS for multiplexed detection [50].
Workflow Diagram:
Step-by-Step Procedure:
Cohort and Sample Preparation:
Immunoaffinity Capture:
- Pool equal volumes of magnetic beads (e.g., Dynabeads M-270) conjugated to monoclonal antibodies against each target protein (e.g., CD5L, APOA4, IBP3) [50].
- Using a robotic liquid handler, transfer the pooled "Ab-beads" to a 96-well plate.
- Add patient plasma samples (e.g., 10 µL), calibrator standards, and quality control (QC) samples (e.g., a reference plasma pool) to the plate [50].
- Incubate the plate at 37°C for 90 minutes with intermittent shaking to facilitate antigen-antibody binding [50].
Wash and Digestion:
- Place the plate on a magnet to capture the beads and remove the supernatant containing unbound proteins.
- Wash the beads with a buffer (e.g., PBS) to remove non-specifically bound materials while the beads are immobilized by the magnet [50].
- With the beads still immobilized, remove the final wash and perform on-bead protein digestion. Add trypsin directly to the beads to digest the captured target proteins, followed by reduction and alkylation steps [50]. This elutes the targets as peptides directly into the solution.
LC-MRM/MS Analysis:
- Transfer the peptide-containing supernatant to a new plate for analysis.
- Utilize a microflow or low-flow LC system coupled to a triple quadrupole MS to drastically reduce chromatography time (e.g., from 90 min to 8 min) while maintaining robust performance [50].
- Program the MRM method to monitor specific transitions for the signature peptides of the three captured proteins and their corresponding SIS peptides.
Data Analysis:
- Interpolate the peak area ratios (light/heavy) of the samples against the calibrator curve to determine the absolute concentration of each biomarker [50].
- Incorporate these protein concentrations along with the patient's clinical variables into a pre-defined algorithm to generate a final risk score (e.g., for predicting DKD) [50].
Application Note 3: Endocrine Testing Biomarker Panels
Context and Rationale
Endocrine testing, which focuses on detecting disorders by measuring hormone levels, is shifting strongly from traditional immunoassays to mass-spectrometry-based assays [4] [3] [52]. While immunoassays have been the historical mainstay, they suffer from significant drawbacks, most notably inadequate specificity due to antibody cross-reactivity, which can lead to false positives and overestimated concentrations [4] [3]. Mass spectrometry offers superior specificity, accuracy, and the critical advantage of being a multi-component method, allowing for steroid profiling—the simultaneous quantification of multiple steroids in a single analysis to investigate entire metabolic pathways [4] [3]. This capability is essential for the complex evaluation of steroidogenesis and has led to the recommendation by the Journal of Clinical Endocrinology and Metabolism to use MS over immunoassays for measuring sex steroids [3].
Exemplary Study and Data
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) with QqQ instruments is now identified as a reference measurement procedure in Clinical Standardization Programs organized by the Centers for Disease Control and Prevention (CDC) [3]. The trend is toward developing methods that cover an increasing part of the "steroidome." For example, a method based on GC–triple quadrupole MS for the determination of a hundred endogenous steroids in human serum has recently been presented [3]. The number of participants in the UK NEQAS steroid hormone proficiency testing scheme using LC-MS/MS increased from 3% to 18% between 2011 and 2019, underscoring its growing adoption [3].
Table 3: Comparison of Immunoassay vs. Mass Spectrometry for Endocrine Testing
| Characteristic | Immunoassay | Triple Quadrupole MS |
|---|---|---|
| Specificity | Prone to cross-reactivity, leading to inaccurate results [3] | High specificity via MS/MS fragmentation; reduced false positives [4] [3] |
| Multiplexing | Generally single-analyte [50] | True multiplexing; simultaneous steroid profiling in one run [4] [3] |
| Accuracy | Can overestimate concentrations; method-dependent variability [3] | High accuracy and precision; considered a reference method [3] |
| Workflow | High-throughput, well-established [50] | Technically complex, but throughput is increasing with automation [47] |
| Primary Application | High-volume routine testing [50] | Reference methods, complex cases, steroid profiling, and when high specificity is required [4] [3] [52] |
Detailed Protocol: Steroid Profiling from Serum by LC-MRM/MS
Principle: This protocol outlines the simultaneous quantification of a panel of steroid hormones (e.g., cortisol, testosterone, aldosterone) from human serum using liquid-liquid extraction followed by LC-MRM/MS. The use of stable isotope-labeled internal standards for each steroid is critical for accurate quantification.
Workflow Diagram:
Step-by-Step Procedure:
Sample Preparation:
- Thaw frozen serum samples on ice or at 4°C.
- Aliquot a known volume of serum (e.g., 100-500 µL) into a glass tube.
- Add a mixture of SIS for each target steroid to the sample. This corrects for losses during sample preparation and ionization variability in the MS [47].
Liquid-Liquid Extraction (LLE):
- Add an organic solvent (e.g., methyl tert-butyl ether, MTBE) to the serum to precipitate proteins and extract the steroids into the organic phase.
- Vortex mix vigorously and then centrifuge to separate the phases.
- Transfer the organic (upper) layer to a new tube.
Sample Concentration:
- Evaporate the organic extract to complete dryness under a gentle stream of nitrogen gas in a warm water bath (e.g., 40°C).
- Reconstitute the dried extract in a small volume of LC-MS starting mobile phase (e.g., water with 0.1% formic acid or a water/methanol mixture) to concentrate the analytes.
LC-MRM/MS Analysis:
- Inject the reconstituted sample onto a reversed-phase UHPLC column (e.g., C18) for separation. A gradient from aqueous to organic mobile phase is used to elute the steroids based on their hydrophobicity.
- The eluent is introduced into the triple quadrupole mass spectrometer equipped with an electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI) source.
- The MS is operated in positive MRM mode, monitoring specific precursor > product ion transitions for each steroid and its corresponding SIS.
Data Analysis:
- Generate a calibration curve for each steroid by analyzing serially diluted calibrators with known concentrations.
- Calculate the peak area ratio (analyte/SIS) for each sample and interpolate the concentration from the linear regression of the calibration curve.
The Scientist's Toolkit: Essential Research Reagents and Materials
The successful development and validation of a clinical MS-based biomarker assay rely on a suite of specialized reagents and materials. The following table details key components of this toolkit.
Table 4: Essential Research Reagent Solutions for Targeted Biomarker Assays
| Reagent / Material | Function / Description | Application Examples |
|---|---|---|
| Stable Isotope-Labeled (SIS) Peptides/Proteins [47] | Internal standards for absolute quantification; correct for analytical variation during sample prep and MS analysis. | SIS peptides added post-digestion; SIS proteins or "winged" peptides added pre-digestion for higher accuracy [47]. |
| Immunoaffinity Depletion Columns (e.g., MARS14) [48] [47] | Remove top 14 abundant plasma proteins (e.g., albumin, IgG) to enhance detection of lower-abundance biomarkers. | Critical for plasma/serum analysis in cancer biomarker discovery and verification [48]. |
| Anti-Peptide Antibodies (Bead-Conjugated) [50] | Monoclonal antibodies immobilized on magnetic beads for specific enrichment of target proteins from complex samples. | Used in high-throughput immunoaffinity-MRM assays like PromarkerD to capture CD5L, APOA4, and IBP3 [50]. |
| Trypsin, Sequencing Grade [48] [50] | Protease enzyme that specifically cleaves proteins at the C-terminal side of lysine and arginine residues, generating peptides for MS analysis. | Standard for "bottom-up" proteomics in nearly all protein biomarker workflows [48]. |
| Liquid Chromatography Columns (C18 reversed-phase) [48] [47] | Separate peptides based on hydrophobicity prior to introduction into the mass spectrometer. | NanoLC or microflow columns for high-sensitivity analysis; UHPLC columns for faster, high-resolution separations [48] [50]. |
| Quality Control (QC) Reference Plasma [50] | A pooled plasma sample from multiple donors, aliquoted and stored at -80°C, used to monitor assay performance and reproducibility across batches. | Run with every batch of clinical samples to ensure precision and stability of the analytical platform [50]. |
Triple quadrupole mass spectrometry, particularly in MRM mode, has firmly established itself as a powerful and versatile platform for the validation of protein biomarker panels across diverse disease areas. Its superior specificity, quantitative accuracy, and ability to multiplex make it an indispensable tool for bridging the gap between biomarker discovery and clinical application. As demonstrated in the application notes for cancer, diabetic kidney disease, and endocrine testing, robust and carefully optimized protocols enable the translation of promising biomarker candidates into clinically actionable tests. The ongoing development of more efficient sample preparation methods, faster chromatography, and increasingly sensitive instrumentation promises to further expand the role of QqQ MS in personalized medicine, ultimately contributing to improved disease detection, monitoring, and patient outcomes.
High-Throughput Quantitative Analysis in Pharmacokinetics and Therapeutic Drug Monitoring
The integration of high-throughput screening (HTS) technologies with quantitative analytical methods has revolutionized modern pharmacokinetics and therapeutic drug monitoring (TDM). This approach enables the simultaneous evaluation of thousands of compounds or patient samples, significantly accelerating drug discovery and personalized treatment strategies [53] [54]. In pharmacokinetics, quantitative HTS (qHTS) allows for the generation of concentration-response data across multiple concentrations for vast chemical libraries, providing robust datasets for structure-activity relationship analysis [53]. For TDM, the application of high-throughput mass spectrometry techniques, particularly liquid chromatography-tandem mass spectrometry (LC-MS/MS), has become the gold standard for measuring drug concentrations in biological fluids with exceptional accuracy, specificity, and sensitivity [55] [56].
The significance of these methodologies extends throughout the drug development pipeline and clinical practice. In drug discovery, high-throughput kinetics enables detailed kinetic characterization of compounds earlier in the process, informing optimization efforts [57]. In clinical settings, LC-MS/MS-based TDM is particularly crucial for drugs with narrow therapeutic windows, such as immunosuppressants, antibiotics, and chemotherapeutic agents, where precise dosing is essential to balance efficacy and toxicity [55] [56]. The continuous advancement in instrumentation, automation, and data analysis workflows has further enhanced the reproducibility and throughput of these quantitative analyses, solidifying their essential role in modern pharmacology and personalized medicine.
Quantitative High-Throughput Screening (qHTS) in Drug Discovery
Fundamentals of qHTS and the Hill Equation
Quantitative high-throughput screening (qHTS) represents a significant evolution from traditional HTS by generating concentration-response data simultaneously for thousands of compounds across multiple concentrations [53]. This approach provides rich datasets for calculating critical pharmacokinetic parameters, notably compound potency (AC50) and efficacy (Emax), which are essential for lead optimization in drug discovery [53]. The methodology employs low-volume cellular systems (e.g., <10 μl per well in 1536-well plates) combined with high-sensitivity detectors, enabling the efficient screening of large chemical libraries with improved false-positive and false-negative rates compared to single-concentration approaches [53].
The Hill equation (HEQN) serves as the fundamental mathematical model for analyzing qHTS data, providing a sigmoidal curve that describes the relationship between compound concentration and biological response [53]. The logistic form of the Hill equation is expressed as:
Where:
- Ri = measured response at concentration Ci
- E0 = baseline response
- E∞ = maximal response
- h = shape parameter (Hill slope)
- AC50 = concentration for half-maximal response
- Ci = compound concentration [53]
Table 1: Key Parameters in Hill Equation Modeling for qHTS
| Parameter | Biological Interpretation | Application in Drug Discovery |
|---|---|---|
| AC50 | Compound potency; concentration producing half-maximal response | Primary parameter for compound ranking and prioritization |
| Emax (E∞ - E0) | Compound efficacy; maximal response achievable | Critical for identifying full agonists vs. partial agonists |
| Hill slope (h) | Steepness of concentration-response curve | Indicator of cooperativity in receptor binding |
| Baseline (E0) | Response in absence of compound | Normalization reference for assay quality control |
Statistical Considerations and Parameter Estimation
Despite the widespread use of the Hill equation, parameter estimation in qHTS presents significant statistical challenges that can impact data reliability [53]. The precision of AC50 and Emax estimates depends heavily on experimental design factors including concentration range selection, response variability, and concentration spacing [53]. Parameter estimation is highly variable when the tested concentration range fails to establish both upper and lower asymptotes of the sigmoidal curve, leading to unreliable potency estimates that can span several orders of magnitude in simulation studies [53].
The impact of experimental replication on parameter estimation precision is substantial, as illustrated in Table 2, which summarizes data from simulation studies of 14-point concentration-response curves. Increasing sample size from n=1 to n=5 significantly narrows the confidence intervals for both AC50 and Emax estimates, particularly for challenging curve shapes where only one asymptote is defined [53]. These findings underscore the importance of adequate replication in qHTS study design to ensure reliable parameter estimation for structure-activity relationship analysis.
Table 2: Impact of Sample Size on Parameter Estimation Precision in Simulated qHTS Data
| True AC50 (μM) | True Emax (%) | Sample Size (n) | AC50 Estimate [95% CI] | Emax Estimate [95% CI] |
|---|---|---|---|---|
| 0.001 | 25 | 1 | 7.92e-05 [4.26e-13, 1.47e+04] | 1.51e+03 [-2.85e+03, 3.10e+03] |
| 0.001 | 25 | 5 | 7.24e-05 [1.13e-09, 4.63] | 26.08 [-16.82, 68.98] |
| 0.001 | 100 | 1 | 1.99e-04 [7.05e-08, 0.56] | 85.92 [-1.16e+03, 1.33e+03] |
| 0.001 | 100 | 5 | 7.24e-04 [4.94e-05, 0.01] | 100.04 [95.53, 104.56] |
| 0.1 | 25 | 1 | 0.09 [1.82e-05, 418.28] | 97.14 [-157.31, 223.48] |
| 0.1 | 25 | 5 | 0.10 [0.05, 0.20] | 24.78 [-4.71, 54.26] |
Mass Spectrometry in Biomarker Discovery and Validation
The Biomarker Development Pipeline
Mass spectrometry-based proteomic analysis has become a cornerstone approach for discovering new disease biomarkers, particularly in oncology [10] [20]. The biomarker development pipeline encompasses multiple preclinical phases before clinical implementation, with an inverse relationship between the number of samples analyzed and the number of proteins quantified at each stage [10]. The initial discovery phase typically employs non-targeted "shotgun" proteomics to identify hundreds of differentially expressed proteins in small sample sets (typically 10-30 samples) [10] [20]. Promising candidates then progress through verification using targeted methods on larger sample sets (50-100 samples), followed by validation on hundreds of samples before ultimately undergoing clinical validation with 500-1000+ samples [10].
This stepwise approach ensures that only the most promising biomarker candidates advance to large-scale clinical testing, optimizing resource allocation [20]. Successful execution requires careful attention to study design, sample collection protocols, statistical power considerations, and analytical validation at each stage [20]. The application of this structured pipeline has led to the identification of numerous potential cancer biomarkers, though only a limited number have received FDA approval to date, highlighting the stringency of the validation process [10].
Mass Spectrometric Methods Across the Pipeline
Different mass spectrometric techniques are employed at various stages of the biomarker development pipeline, each optimized for specific requirements of throughput, multiplexing capacity, and analytical performance [10]. Discovery-phase experiments typically utilize non-targeted approaches with relative quantification, including:
- Isobaric tagging methods (e.g., iTRAQ, TMT)
- Label-free quantification techniques (e.g., spectral counting, peak intensity)
- Gel-based separation combined with MS identification [10]
For verification and validation phases, targeted mass spectrometry approaches, particularly multiple reaction monitoring (MRM) or selected reaction monitoring (SRM), are preferred for their superior sensitivity, specificity, and quantitative precision [10]. These targeted methods focus on specific proteotypic peptides that act as surrogates for the proteins of interest, enabling highly precise quantification of biomarker candidates across large sample cohorts [10] [20]. The transition from discovery to verification represents a critical bottleneck in biomarker development, where MRM-based methods have demonstrated particular utility in bridging these phases [10].
Diagram 1: Biomarker Development Pipeline. This workflow illustrates the sequential stages from discovery to clinical application, showing the inverse relationship between number of proteins analyzed and sample size at each phase.
Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) in Therapeutic Drug Monitoring
Analytical Principles and Applications
Liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS) has emerged as the gold standard methodology for therapeutic drug monitoring (TDM) due to its exceptional specificity, sensitivity, and multiplexing capabilities [55] [56]. The technique combines the separation power of liquid chromatography with the detection specificity of tandem mass spectrometry, enabling precise quantification of drugs and metabolites in complex biological matrices such as blood, plasma, and serum [55]. This analytical approach is particularly valuable for TDM of drugs with narrow therapeutic indices, where accurate measurement is essential for optimizing efficacy while minimizing toxicity [56].
LC-MS/MS has become the reference method for monitoring immunosuppressive drugs (e.g., tacrolimus, cyclosporine, sirolimus) in transplant patients, where maintaining concentrations within a narrow therapeutic range is critical for preventing organ rejection while avoiding drug-related toxicities [56]. The technology has also been widely applied for TDM of glycopeptide antimicrobials (e.g., vancomycin, teicoplanin), where pharmacokinetic/pharmacodynamic (PK/PD) properties guide dosing strategies to maximize antibacterial efficacy [55]. The superior performance of LC-MS/MS compared to immunoassays includes reduced metabolite interference, lower cross-reactivity concerns, and the ability to simultaneously measure multiple analytes, making it indispensable for modern TDM applications [56].
Advantages Over Alternative Methodologies
The dominance of LC-MS/MS in TDM applications stems from several distinct advantages over traditional immunoassay techniques [56]. Specificity is significantly enhanced due to the dual separation mechanisms of LC (chromatographic retention time) and MS/MS (precursor ion → fragment ion transitions), virtually eliminating interference from structurally similar compounds or metabolites [56]. Studies have demonstrated that immunoassays for immunosuppressant drugs can show positive biases of up to 20% compared to LC-MS/MS due to metabolite cross-reactivity [56]. Similarly, sensitivity limitations of immunoassays are overcome by LC-MS/MS, which can reliably quantify drugs at low picogram per milliliter levels, essential for drugs with sub-therapeutic concentrations in the nanogram per milliliter range [20] [56].
The multiplexing capability of LC-MS/MS allows simultaneous quantification of multiple drugs and their metabolites in a single analytical run, providing comprehensive pharmacokinetic profiles from minimal sample volume [55] [56]. This feature is particularly valuable in transplant medicine, where patients typically receive combination immunosuppressant therapy [56]. Additionally, LC-MS/MS methods offer greater flexibility for method development and refinement compared to commercially available immunoassays, though this advantage must be balanced against challenges in standardization and interlaboratory reproducibility [56].
Diagram 2: LC-MS/MS Workflow for TDM. The process illustrates the sequential steps from sample preparation to detection, highlighting the key components of the triple quadrupole mass spectrometer commonly used for quantitative analysis.
Experimental Protocols
Protocol: LC-MS/MS Method for Glycopeptide Antimicrobial TDM
Principle: This protocol describes a validated LC-MS/MS method for simultaneous quantification of glycopeptide antimicrobials (vancomycin, teicoplanin, dalbavancin, oritavancin, telavancin) in human plasma for TDM applications [55].
Materials and Reagents:
- Analytical standards: Vancomycin, teicoplanin, dalbavancin, oritavancin, telavancin
- Internal standards: Stable isotope-labeled analogs of each analyte
- Mobile phases: LC-MS grade water with 0.1% formic acid; LC-MS grade methanol with 0.1% formic acid
- Sample preparation: Protein precipitation reagents (acetonitrile, methanol)
- Calibrators and quality controls: Prepared in drug-free human plasma
Sample Preparation:
- Aliquot 100 μL of patient plasma into microcentrifuge tubes
- Add 20 μL of internal standard working solution to each sample
- Precipitate proteins by adding 300 μL of ice-cold acetonitrile
- Vortex mix for 30 seconds, then centrifuge at 14,000 × g for 10 minutes
- Transfer 200 μL of supernatant to autosampler vials for analysis
LC-MS/MS Conditions:
- Chromatography: Reverse-phase C18 column (100 × 2.1 mm, 2.6 μm)
- Mobile phase gradient: 10-95% methanol over 5 minutes
- Flow rate: 0.3 mL/min
- Injection volume: 5 μL
- Ionization: Electrospray ionization (ESI) in positive mode
- Mass transitions: Monitor 2-3 multiple reaction monitoring (MRM) transitions per analyte
Quantification:
- Generate calibration curves using peak area ratios (analyte/internal standard) versus concentration
- Use weighted (1/x²) linear regression for curve fitting
- Apply acceptance criteria: calibration standards within ±15% of nominal values (±20% at LLOQ)
- Calculate patient sample concentrations using regression equation
Protocol: Quantitative HTS Using Concentration-Response Screening
Principle: This protocol outlines a qHTS approach for generating concentration-response curves for large compound libraries, enabling simultaneous assessment of potency and efficacy [53].
Materials and Reagents:
- Compound library: Dissolved in DMSO at 10 mM stock concentration
- Assay plates: 1536-well microtiter plates
- Cell culture: Appropriate cell line for target pathway
- Detection reagents: Fluorescent or luminescent readout compatible with HTS
- Automated liquid handling systems: For compound and reagent addition
Procedure:
- Plate design: Implement standardized plate layouts with positive (100% effect) and negative (basal) controls
- Compound transfer: Using automated liquid handlers, transfer compound stocks to assay plates across a range of concentrations (typically 8-15 points with 1:3 or 1:2 serial dilutions)
- Cell seeding: Add cell suspension to assay plates (typically <10 μL per well in 1536-well format)
- Incubation: Incubate plates under appropriate conditions (time, temperature, CO₂)
- Signal detection: Add detection reagents and measure response using plate readers
- Data capture: Export raw data to specialized HTS analysis software
Data Analysis:
- Normalization: Convert raw signals to percentage activity relative to controls
- Curve fitting: Fit normalized data to Hill equation using nonlinear regression
- Parameter estimation: Calculate AC50, Emax, and Hill slope for each compound
- Quality assessment: Apply quality control criteria based on control performance and curve fit statistics
Troubleshooting:
- Poor curve fits: Ensure concentration range adequately defines both asymptotes
- High variability: Include replicate measurements and assess edge effects
- Systematic error: Implement randomization schemes to minimize batch effects
Research Reagent Solutions
Table 3: Essential Research Reagents for High-Throughput Quantitative Analysis
| Reagent Category | Specific Examples | Function and Application |
|---|---|---|
| Mass Spectrometry Standards | Stable isotope-labeled internal standards (e.g., ¹³C, ¹⁵N analogs), purified analyte standards | Enable precise quantification via isotope dilution mass spectrometry; essential for LC-MS/MS method development and validation [55] [20] |
| Chromatography Materials | LC-MS grade solvents (water, methanol, acetonitrile), volatile modifiers (formic acid, ammonium acetate), reverse-phase columns (C18, C8) | Provide optimal separation efficiency and ionization efficiency for LC-MS/MS analysis; critical for resolving analytes from matrix components [55] [56] |
| HTS Compound Libraries | Diverse small molecule collections, targeted chemotypes, natural product extracts | Source of chemical matter for screening campaigns; foundation for structure-activity relationship studies [53] [54] |
| Cell-Based Assay Reagents | Reporter gene systems, fluorescent probes, viability indicators, growth media | Enable biological activity assessment in high-throughput format; crucial for functional characterization of compounds [54] [57] |
| Sample Preparation Kits | Protein precipitation plates, solid-phase extraction cartridges, supported liquid extraction devices | Facilitate efficient cleanup of biological samples; improve assay sensitivity and specificity while maintaining throughput [55] [56] |
| Quality Control Materials | Certified reference materials, quality control pools at multiple concentrations, matrix-matched calibrators | Ensure analytical method reliability and longitudinal data comparability; essential for method validation and implementation [20] [56] |
Maximizing Assay Performance: Strategies to Overcome Sensitivity and Specificity Challenges
Identifying and Mitigating Ion Suppression in Complex Biological Samples
Ion suppression is a pervasive matrix effect in mass spectrometry that significantly compromises the sensitivity, accuracy, and precision of quantitative bioanalysis, particularly in biomarker validation using triple quadrupole mass spectrometry. This phenomenon occurs when co-eluting matrix components interfere with the ionization efficiency of target analytes, leading to reduced signal intensity and unreliable quantification [58] [59]. As regulatory expectations for sensitivity and reproducibility in drug development continue to rise, implementing robust strategies to overcome ion suppression has become imperative for generating credible analytical data [58]. This application note provides detailed protocols and methodologies for identifying, quantifying, and mitigating ion suppression within the context of biomarker research.
Understanding Ion Suppression: Mechanisms and Impact
Ion suppression primarily stems from competition during the ionization process. In electrospray ionization (ESI), co-eluting substances can impede the efficient transfer of target analytes from solution to the gas phase through several mechanisms: by altering droplet formation and solvent evaporation rates, by competing directly for available charge, or through gas-phase proton transfer reactions that favor matrix components over the analytes of interest [58] [59].
The consequences of unaddressed ion suppression are severe for biomarker validation workflows. It can cause:
- Decreased sensitivity, elevating limits of detection and potentially obscuring low-abundance biomarkers [58]
- Reduced accuracy and precision, leading to inaccurate concentration measurements that misrepresent biological reality [60]
- Compromised data quality, introducing variability that undermines the reliability of biomarker assays [58] [60]
Recent research has demonstrated that ion suppression affects nearly all metabolites in liquid chromatography-mass spectrometry (LC-MS) analyses, with suppression effects ranging from 1% to over 90% across different chromatographic systems and biological matrices [60]. This extensive variability poses a fundamental challenge for biomarker validation, where consistent and reproducible quantification is paramount.
Systematic Strategies for Mitigating Ion Suppression
A multi-faceted approach is essential for effectively overcoming ion suppression in complex biological samples. The following strategies can be implemented individually or in combination to enhance data quality.
Sample Preparation Techniques
- Solid-Phase Extraction (SPE): Effectively removes proteins, phospholipids, and other endogenous interferents from biological samples like plasma and serum. Selective SPE sorbents can target specific compound classes while eliminating matrix components responsible for suppression [58].
- Protein Precipitation: While simple and rapid, this method may leave behind significant matrix components that cause ion suppression. It is often most effective when combined with additional cleanup steps or chromatographic optimization [58].
- Sample Dilution: Reduces the overall matrix concentration but may compromise sensitivity for low-abundance biomarkers. The optimal dilution factor must balance sufficient detection capability with acceptable suppression levels [60].
Chromatographic Optimization
Chromatographic separation represents one of the most powerful approaches for mitigating ion suppression by temporally separating analytes from matrix interferents.
Table 1: Chromatographic Approaches to Reduce Ion Suppression
| Approach | Mechanism | Application Considerations |
|---|---|---|
| Improved Peak Resolution | Increases separation between analytes and matrix components | Utilize longer columns, smaller particle sizes, or optimized gradients [58] |
| Microflow LC | Reduces initial droplet size in ESI, minimizing co-elution | Can provide up to 6-fold sensitivity improvement [58] |
| Column Chemistry Selection | Tailors selectivity to separate analytes from specific matrix interferents | HILIC, RPLC, and IC each offer distinct separation mechanisms [60] [61] |
| Retention Time Shift | Moves analytes away from regions of high matrix interference | Adjust mobile phase pH, solvent strength, or gradient profile [58] |
Mass Spectrometric and Source Optimization
Instrument parameter optimization can significantly reduce susceptibility to ion suppression effects:
- Source Geometry and Positioning: Proper alignment of the ionization source relative to the MS inlet ensures efficient ion transmission and minimizes contamination buildup [58].
- Gas Flow and Temperature Optimization: Fine-tuning desolvation gas flow rates and temperatures enhances droplet desolvation efficiency, reducing the opportunity for competitive ionization [58].
- Reduced Pressure Ionization: Emerging techniques utilizing sub-ambient pressure around the ESI emitter have demonstrated 7- to 20-fold signal enhancements in high-salt solutions by producing smaller initial droplets and improving desolvation [62].
- Interface Parameter Tuning: Capillary voltage and nebulizing gas pressure significantly impact ionization efficiency and should be optimized for each analyte class [58].
Advanced Chemical and Computational Approaches
- Stable Isotope-Labeled Internal Standards (SIL-IS): Effectively correct for variability in ionization efficiency when added prior to sample preparation. They account for analyte-specific suppression by exhibiting identical chemical behavior to their native counterparts [60].
- Chemical Isotope Labeling (CIL): Uses targeted derivatization with light and heavy isotope tags to enhance ionization efficiency and enable accurate quantification through peak-pair detection [61].
- IROA TruQuant Workflow: An advanced method employing a stable isotope-labeled internal standard library with companion algorithms to measure and correct for ion suppression across all detected metabolites in non-targeted studies [60].
The following workflow diagram illustrates a comprehensive approach to identifying and mitigating ion suppression in biomarker research:
Experimental Protocols for Ion Suppression Assessment
Post-Column Infusion Analysis for Suppression Zone Mapping
This protocol visually identifies chromatographic regions where ion suppression occurs by monitoring a constant analyte infusion during a matrix blank injection.
Materials:
- Triple quadrupole mass spectrometer with LC system
- Analytical column appropriate for target analytes
- Stock solution of test analyte (e.g., biomarker standard)
- Matrix blanks (e.g., stripped plasma, surrogate matrix)
Procedure:
- Prepare a working solution of the test analyte at a concentration that produces a stable baseline signal.
- Connect a tee-fitting between the LC column outlet and the MS inlet.
- Infuse the test analyte solution post-column at a constant flow rate (e.g., 5-10 μL/min) using a syringe pump.
- Inject a processed matrix blank sample and run the chromatographic method.
- Monitor the MRM transition for the test analyte throughout the chromatographic run.
Interpretation: Regions where the analyte signal decreases (dips in the baseline) indicate the elution of matrix components that cause ion suppression. These "suppression zones" should be noted, and method development should focus on shifting target analyte retention times away from these regions [58].
Quantitative Ion Suppression Assessment Using Standard Addition
This method quantifies the magnitude of ion suppression for specific analytes by comparing responses in clean versus matrix-containing samples.
Materials:
- Matrix-matched calibration standards
- Solvent-based calibration standards
- Quality control samples at low, medium, and high concentrations
Procedure:
- Prepare two sets of calibration standards: one in processed biological matrix and another in pure solvent, both covering the same concentration range.
- Prepare quality control samples in matrix at three concentration levels.
- Analyze all samples using the LC-MS/MS method.
- Compare the slope of the matrix-matched calibration curve to the solvent-based calibration curve.
- Calculate the ion suppression factor (ISF) using the formula:
Calculation: ISF = (Slopematrix / Slopesolvent) × 100%
Interpretation: ISF values below 85% or above 115% typically indicate significant ion suppression or enhancement, respectively, requiring method modification [58] [60].
IROA TruQuant Workflow for Comprehensive Suppression Correction
The IROA (Isotopic Ratio Outlier Analysis) TruQuant workflow provides a sophisticated solution for measuring and correcting ion suppression across all detected metabolites in non-targeted studies.
Table 2: IROA TruQuant Workflow Components
| Component | Composition | Role in Suppression Correction |
|---|---|---|
| IROA Internal Standard (IROA-IS) | 95% (^{13})C-labeled metabolite library | Spiked into each sample at constant concentration to measure suppression [60] |
| IROA Long-Term Reference Standard (IROA-LTRS) | 1:1 mixture of 95% (^{13})C and 5% (^{13})C standards | Provides reference isotopic patterns for peak identification [60] |
| ClusterFinder Software | Proprietary algorithm (v4.2.21+) | Automatically calculates and corrects ion suppression using Eq. 1 [60] |
| Dual MSTUS Normalization | MS Total Useful Signal algorithm | Normalizes data based on useful signal across samples [60] |
Procedure:
- Spike IROA-IS into all samples during initial preparation.
- Add IROA-LTRS to establish reference isotopic patterns.
- Process samples using appropriate LC-MS method (IC, HILIC, or RPLC in positive/negative mode).
- Analyze data using ClusterFinder software, which identifies metabolites based on their characteristic IROA isotopolog ladders.
- Apply the suppression correction equation:
Calculation: [AUC\text{-}12C{\text{suppression-corrected}} = AUC\text{-}12C{\text{observed}} \times \frac{AUC\text{-}13C{\text{expected}}}{AUC\text{-}13C{\text{observed}}}] Where AUC-13C_expected is the constant value determined from the IROA-IS [60].
Validation: This workflow has demonstrated effective correction of ion suppression ranging from 1% to >90% across diverse analytical conditions, restoring expected linear response with sample input volume [60].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagents for Ion Suppression Mitigation
| Reagent / Material | Function | Application Notes |
|---|---|---|
| Stable Isotope-Labeled Internal Standards | Correct for analyte-specific suppression; account for recovery variations | Should be added as early as possible in sample preparation [60] |
| IROA Internal Standard Library | Comprehensive suppression correction for non-targeted metabolomics | Enables measurement and correction of ion suppression across all detected metabolites [60] |
| Chemical Isotope Labeling Kits (CIL-4101-KT, CIL-4145-KT) | Enhance detection sensitivity and quantification accuracy | Use dansyl chloride for amine/phenol and hydroxyl submetabolomes [61] |
| Hybrid Surface LC Columns | Minimize analyte adsorption and peak tailing | Reduce secondary interactions that contribute to suppression [58] |
| Volatile Mobile Phase Additives | Promote efficient droplet desolvation | Ammonium formate/acetate preferred over non-volatile salts [58] |
| Reduced Pressure Ionization Chamber | Enhance signal in high-salt conditions | Custom 3D-printed chambers can interface with commercial MS systems [62] |
Effective management of ion suppression is not merely an optimization step but a fundamental requirement for robust biomarker validation using triple quadrupole mass spectrometry. By implementing the systematic assessment protocols and mitigation strategies outlined in this application note—including appropriate sample cleanup, chromatographic optimization, advanced isotope labeling techniques, and instrumental parameter tuning—researchers can significantly improve data quality and reliability. The integration of innovative approaches like the IROA TruQuant workflow and reduced pressure ionization provides powerful new tools to overcome the challenges posed by complex biological matrices, ultimately supporting more confident biomarker discovery and validation in drug development pipelines.
In the field of biomarker validation using triple quadrupole mass spectrometry, the strategic application of mass resolution is a critical factor for achieving the requisite selectivity, particularly when analyzing complex biological matrices. The biomarker validation pipeline is a multi-stage process, progressing from discovery to verification and, finally, to clinical validation [10]. In the verification phase, selected reaction monitoring (SRM) on triple quadrupole (QQQ) mass spectrometers is the method of choice for its superior sensitivity and quantitative capabilities [24]. The core of an SRM experiment involves selecting a precursor ion in the first quadrupole (Q1) and a specific fragment ion in the third quadrupole (Q3) [24]. The resolution settings applied in these mass filters are paramount for isolating the target ions from chemical background and co-eluting interferences, directly impacting the reliability of the quantitative data. This document outlines detailed protocols and application notes for leveraging resolution settings to enhance selectivity in SRM-based biomarker assays.
Technical Background: Resolution in the Biomarker Validation Pipeline
Mass resolution, conventionally defined as the smallest separation between two peaks of equal height and width that still shows a valley, is a key parameter in mass analysis [63]. In practice, the required mass resolving power is significantly higher when peaks are of unequal height, as is common in biological samples where high dynamic range is the norm [63].
The Role of SRM in Biomarker Verification
The journey of a protein biomarker from discovery to clinical application is long and fraught with challenges. Discovery experiments, often using non-targeted "shotgun" proteomics, typically yield a long list of candidate proteins that are differentially expressed between healthy and diseased states [10]. The subsequent verification phase narrows this list by quantifying candidate biomarkers in a larger set of patient samples (e.g., 10-50). It is at this critical juncture that targeted MS approaches, specifically SRM on a QQQ mass spectrometer, come to the fore [10] [20]. SRM's power lies in its two stages of mass filtering, which can increase sensitivity by one to two orders of magnitude compared to full-scan methods, making it ideal for quantifying low-abundance biomarkers in complex mixtures like plasma or serum [24].
Table: Key Stages in the Biomarker Development Pipeline
| Stage | Objective | Typical Sample Number | Primary MS Method | Data Output |
|---|---|---|---|---|
| Discovery | Identify candidate biomarkers | Small | Non-targeted (Shotgun) Proteomics | "Up-or-down" regulation, fold changes [10] |
| Verification | Verify candidates on larger sample sets | 10-50 | Targeted MS (e.g., SRM/MRM) | Quantitative data on selected proteins [10] [20] |
| Validation | Clinically validate final biomarkers | 100-1000s | Immunoassays, targeted MS | Protein concentrations for clinical use [10] |
Application Note: Strategic Resolution Modes for Selectivity
The balance between unit mass resolution and enhanced resolution (<0.1 Da) in Q1 and Q3 can be strategically manipulated to optimize an assay's selectivity, sensitivity, and speed.
Unit Mass Resolution: Maximum Sensitivity
Operating at unit mass resolution (a peak width of ~0.7 Da at full width half maximum) allows the entire isotopic envelope of a peptide precursor ion to pass through the quadrupole mass filter. This maximizes ion transmission and is the preferred setting for achieving the highest sensitivity, which is often essential for detecting low-abundance biomarkers. However, this mode is more susceptible to interference from isobaric compounds that share a nominal mass but have a different exact mass (mass defect) [63].
Enhanced Resolution (<0.1 Da): Maximum Selectivity
Applying enhanced resolution (narrowing the peak width to <0.1 Da) allows the quadrupole to isolate a specific isotope of the target ion (typically the monoisotopic peak) based on its exact mass. This significantly improves selectivity by resolving the target from potential isobaric interferences that have a slightly different exact mass. The trade-off is a reduction in ion transmission and thus sensitivity, as a narrower mass window is being transmitted [63].
Table: Comparison of Unit Mass and Enhanced Resolution Modes in SRM
| Parameter | Unit Mass Resolution | Enhanced Resolution (<0.1 Da) |
|---|---|---|
| Definition | Peak width of ~0.7 Da (FWHM) | Peak width of <0.1 Da (FWHM) |
| Ions Transmitted | Entire isotopic envelope | Primarily the monoisotopic peak |
| Primary Advantage | Maximum sensitivity | Maximum selectivity |
| Primary Disadvantage | Susceptible to isobaric interference | Reduced sensitivity |
| Ideal Use Case | Samples with simple matrix or high-abundance analytes | Complex matrices (e.g., plasma, urine) or low-abundance analytes with interferences |
Experimental Protocol: A Workflow for Method Development
This protocol provides a step-by-step guide for developing a robust SRM assay for biomarker verification, with a focus on optimizing resolution for selectivity.
Step 1: Transition Selection and Peptide Optimization
- Select Proteotypic Peptides: For each candidate biomarker protein, select 2-3 proteotypic peptides (PTPs) that are unique to the protein, have good ionization efficiency, and are within the mass range of the instrument [24].
- Define SRM Transitions: For each peptide, select 2-4 optimal fragment ions.
y-type ions are often predominant and stable in QQQ systems [24]. Heuristic: Choose fragment ions with m/z larger than the precursor m/z to reduce chemical background [24].
Step 2: Initial Method Scouting with Unit Resolution
- Initial Setup: Configure the QQQ instrument for standard unit mass resolution in both Q1 and Q3 (e.g., 0.7 Da FWHM).
- Data Acquisition: Inject a representative matrix sample spiked with the target analyte (or a heavy isotope-labeled version) and acquire SRM data for all predefined transitions.
- Assessment: Review chromatograms for peak shape, signal intensity, and potential co-elution. This establishes a sensitivity baseline.
Step 3: Systematic Resolution Optimization
- Identify Candidates for Enhanced Resolution: For transitions showing poor peak shape, high background, or suspected interferences in the unit mass mode, proceed to optimization.
- Iterative Narrowing: For the affected transitions, systematically narrow the resolution setting in Q1 (e.g., from 0.7 Da to 0.5 Da, 0.3 Da, 0.1 Da) while monitoring the signal-to-noise (S/N) ratio of the target peak.
- S/N increases: Indicates that interfering ions are being excluded. Continue narrowing until S/N plateaus or begins to drop.
- S/N decreases sharply: Indicates the target ion is being lost. Widen the resolution to the previous setting.
- Final Method: Establish the optimal resolution width for each transition that provides the best balance of selectivity and sensitivity. A final method will often use a hybrid of unit mass and enhanced resolution settings for different transitions.
The following workflow diagram summarizes the logical process of method development and optimization.
The Scientist's Toolkit: Essential Reagents and Materials
The following table lists key reagents and materials crucial for successfully executing the SRM-based biomarker validation workflow described in this document.
Table: Essential Research Reagent Solutions for SRM Biomarker Validation
| Item | Function/Application | Key Considerations |
|---|---|---|
| Heavy Isotope-Labeled Peptides | Internal standards for precise quantification; used to distinguish target peptides from unspecific signals and correct for sample preparation variability [24]. | Should be identical in sequence to the target proteotypic peptide, but contain heavy isotopes (e.g., 13C, 15N). |
| Trypsin (Sequencing Grade) | Proteolytic enzyme for digesting proteins into peptides for LC-SRM-MS analysis. | High purity and specificity are required to ensure reproducible and complete digestion. |
| Stable Isotope Labeling with Amino Acids in Cell Culture (SILAC) | Metabolic labeling strategy for relative quantitation in discovery phases that can inform SRM assay development [10]. | Allows for precise comparison of protein abundance between different cell states. |
| Sample Preparation Kits & Reagents | For sample cleanup, depletion of high-abundance proteins (e.g., from plasma), and peptide purification to reduce sample complexity. | Reduces matrix effects and ion suppression, improving SRM assay performance [20]. |
| LC-MS Grade Solvents | Mobile phases for liquid chromatography (e.g., water, acetonitrile) with additives like formic acid. | High purity is essential to minimize chemical background noise and maintain instrument performance. |
Data Interpretation and Analysis
The ultimate goal of resolution optimization is to generate high-quality quantitative data. After data acquisition, careful analysis is required.
- Chromatographic Integration: Integrate the peak areas for both the native analyte and the heavy isotope-labeled internal standard for each transition.
- Peak Shape and Consistency: Examine peak shapes across all samples. A sudden change in shape or retention time in a subset of samples may indicate a persistent interference not fully resolved by the chosen method.
- Ratio and Precision: Calculate the ratio of the native to internal standard peak areas. Assess the precision (e.g., % coefficient of variation) of these ratios across technical and biological replicates. High precision indicates a robust assay.
- Statistical Analysis: Perform appropriate statistical tests to determine if the quantified levels of the candidate biomarker are significantly different between the case and control groups, thereby verifying its potential clinical utility [20].
The strategic balance between unit mass and enhanced resolution is a powerful tool in the mass spectrometrist's arsenal for developing selective SRM assays. By following a systematic method development workflow that includes initial scouting with unit resolution and subsequent application of enhanced resolution to problematic transitions, researchers can significantly improve the selectivity of their biomarker verification assays. This rigorous approach to method development is fundamental to generating reliable data, ultimately accelerating the translation of promising biomarkers from the bench to the clinic.
Optimizing Collision Energies and Ion Source Parameters
In the rigorous field of biomarker validation, achieving high levels of specificity, sensitivity, and reproducibility is paramount. Liquid chromatography coupled with triple quadrupole mass spectrometry (LC-QqQ-MS) has become the cornerstone technique for quantitative bioanalysis in drug development and clinical diagnostics due to its exceptional selectivity and sensitivity in Multiple Reaction Monitoring (MRM) mode [64] [3]. The performance of these methods is critically dependent on the precise optimization of key mass spectrometric parameters, primarily collision energies (CE) and ion source settings. Proper tuning ensures that the instrument operates at its peak, enabling the reliable detection and quantification of biomarkers at low concentrations in complex biological matrices, which is a fundamental requirement for successful biomarker validation studies [65] [58]. This document provides detailed application notes and protocols for systematically optimizing these parameters within the context of a biomarker validation pipeline.
Key Parameters for Optimization in Triple Quadrupole Systems
The performance of a triple quadrupole mass spectrometer is governed by a set of interdependent parameters that control the ionization, transmission, and fragmentation of analyte ions.
Ion Source Parameters
The ion source is where gas-phase ions are generated from the liquid chromatographic effluent. Its parameters are crucial for maximizing ionization efficiency and ensuring stable ion generation [65] [58].
- GS1 (Nebulizer Gas): This gas shears the liquid stream into fine droplets, assisting in droplet formation. Optimal setting promotes efficient desolvation [65].
- GS2 (Heating Gas) and TEM (Source Temperature): The heated gas flow (GS2) and its associated temperature (TEM) facilitate the evaporation of solvent from the charged droplets, promoting the release of gas-phase ions. Higher temperatures generally aid desolvation but must be balanced against the potential for thermal degradation of the analyte [65].
- IS (IonSpray Voltage) or Capillary Voltage: This high voltage applied between the needle and the orifice creates a strong electric field, which is responsible for charging the droplets and ultimately ejecting gas-phase ions into the mass spectrometer [65] [66].
- CUR (Curtain Gas): An inert gas flow that acts as a barrier between the ion source and the orifice to the vacuum system. It helps prevent neutral contaminants and solvent clusters from entering the analyzer, thereby reducing noise and source contamination. This parameter should be maintained as high as possible without a significant loss of sensitivity [65].
Mass Spectrometer Tune Parameters
These parameters control the behavior of ions after they enter the high-vacuum region of the mass spectrometer, influencing fragmentation and transmission [65] [64].
- DP (Declustering Potential): The voltage applied to the orifice to decluster solvated ions by accelerating them, causing collisions with gas molecules that strip away solvent adducts. An excessively high DP can induce unwanted in-source fragmentation, while a low value may result in insufficient declustering [65].
- EP (Entrance Potential): A small potential difference that guides and focuses ions from the interface region into the first quadrupole (Q1). It typically has a minor effect on overall sensitivity [65].
- CE (Collision Energy): The potential difference that accelerates precursor ions selected in Q1 into the collision cell (Q2). The kinetic energy gained by the ions is dissipated through collisions with the collision gas (e.g., nitrogen or argon), leading to fragmentation into product ions. This is one of the most critical parameters to optimize for maximizing the signal of the target product ion [65] [64] [67].
- CAD (Collision Gas): The inert gas introduced into the collision cell (Q2) to enable Collision-Induced Dissociation (CID) of the precursor ions [65] [64].
- CXP (Collision Cell Exit Potential): The voltage applied to guide the product ions out of the collision cell (Q2) and into the third quadrupole (Q3) for mass analysis [65].
Table 1: Key Ion Source and MS Parameters for Optimization
| Parameter | Symbol | Function | Typical Range/Considerations |
|---|---|---|---|
| Nebulizer Gas | GS1 | Assists in droplet formation from LC effluent [65] | Instrument and flow-rate dependent |
| Heating Gas | GS2 | Heated gas that promotes droplet desolvation [65] | Paired with source temperature |
| Source Temperature | TEM | Temperature of the desolvation gas [65] | Balance sensitivity and analyte stability |
| IonSpray Voltage | IS | Creates charged droplets and emits ions [65] | Polarity and instrument dependent |
| Curtain Gas | CUR | Protects orifice from contamination; repulses neutrals [65] | Set as high as possible without losing sensitivity |
| Declustering Potential | DP | Removes solvent clusters from ions [65] | 20 - 100 V; too high can cause fragmentation |
| Entrance Potential | EP | Guides ions into the first quadrupole (Q1) [65] | ~10 V; minor effect on optimization |
| Collision Energy | CE | Energy for fragmenting precursor ions in Q2 [65] [67] | Critical for product ion signal; compound-specific |
| Collision Gas | CAD | Inert gas for collision-induced dissociation [65] | Nitrogen or Argon; set to manufacturer's recommendation |
| Collision Cell Exit Potential | CXP | Transmits product ions from Q2 to Q3 [65] | Optimize for final transmission |
Experimental Protocols for Parameter Optimization
A systematic approach to method development is essential for achieving robust and sensitive bioanalytical methods for biomarker validation.
The first step involves introducing the analyte into the mass spectrometer to begin the optimization process. Two primary techniques are employed [65]:
- Infusion: A purified standard of the analyte (typical concentration 10-500 ng/mL) is continuously introduced into the ion source at a low flow rate (5-25 µL/min) using a syringe pump. This provides a constant signal, allowing for real-time adjustment of parameters without interference from chromatographic effects [65].
- Flow Injection Analysis (FIA): The autosampler injects the analyte standard into a mobile phase stream delivered by the LC pump, which flows directly into the ion source without a chromatographic column. This approach uses the LC system and is typically run at higher flow rates (25-1000 µL/min). It is more amenable to high-throughput optimization [65].
Table 2: Comparison of Sample Introduction Methods
| Method | Required Devices | Typical Flow Rate | Key Advantage |
|---|---|---|---|
| Infusion | Syringe Pump | 5 - 25 µL/min | Continuous signal for real-time tuning |
| Flow Injection Analysis (FIA) | LC Pump & Autosampler | 25 - 1000 µL/min | Higher throughput; uses existing LC system |
Step-by-Step Tuning Procedure
The following protocol outlines a systematic procedure for tuning a triple quadrupole mass spectrometer, adaptable for both manual and automated (e.g., using software like Skyline) optimization workflows [65] [67].
Protocol 1: Systematic Tuning of a Triple Quadrupole Mass Spectrometer
Objective: To optimize ion source and collision cell parameters for a target biomarker analyte. Materials: Purified analyte standard, appropriate solvent (e.g., 50:50 water:acetonitrile with 0.1% formic acid), syringe pump or LC system, triple quadrupole mass spectrometer.
Q1 Scan (Precursor Ion Identification):
- Introduce the analyte standard via infusion or FIA.
- Set the mass spectrometer to scan a relevant m/z range (e.g., 50-1000 Da) in Q1 mode to identify the precursor ion ([M+H]⁺ or [M-H]⁻) of the target compound.
- Once the precursor m/z is confirmed, optimize the Declustering Potential (DP) and Entrance Potential (EP) to maximize the intensity of the precursor ion signal. A typical starting point for EP is ±10 V.
Product Ion Scan (Fragmentation Optimization):
- Set Q1 to transmit only the m/z of the identified precursor ion.
- Set Q3 to scan for product ions over a defined m/z range.
- With the Collision Gas (CAD) set to a medium value, ramp the Collision Energy (CE). The product ion spectrum will reveal the major fragment ions.
- Select the most abundant and specific product ion(s) for the final MRM transition.
MRM Optimization for DP, CE, and CXP:
- Switch the instrument to MRM mode, monitoring the transition from the precursor ion to the selected product ion(s).
- Systemically vary and optimize the CE to achieve the maximum signal for the product ion. This can be done for multiple transitions in parallel.
- Fine-tune the DP and Collision Cell Exit Potential (CXP) for the specific transition to ensure optimal transmission and sensitivity.
Ion Source and Gas Optimization:
- Using the optimized MRM transition, systematically adjust the ion source parameters (GS1, GS2, TEM, IS, CUR) to maximize the signal intensity.
- A "Design of Experiments" (DoE) approach is highly recommended for this step to efficiently explore the multi-dimensional parameter space and identify interactions between factors [66]. For example, a Plackett-Burman design can first screen for significant factors, followed by a response surface methodology (e.g., Central Composite Design) to find the true optimum.
Diagram 1: MS Parameter Optimization Workflow
Advanced and Automated Optimization Strategies
For large-scale biomarker validation studies targeting hundreds of analytes, manual optimization of each parameter becomes impractical. The following advanced strategies are employed:
Prediction of Collision Energy: The optimal CE for a peptide can be predicted using a linear equation of the form
CE = k * (precursor m/z) + b, where the slope (k) and intercept (b) are determined empirically for a given instrument and charge state [67]. This approach, integrated into software like Skyline, allows for the development of sensitive methods without the need for pure standards for every single analyte. Studies have shown that while empirical optimization provides the absolute best signal, predicted CE values can deliver performance within ~8% of the optimum, which is often sufficient for discovery-phase experiments [67].Data-Driven Optimization (DO-MS): This open-source platform uses data from search engines (e.g., MaxQuant) to interactively visualize performance metrics across all levels of the LC-MS/MS analysis [68]. It helps diagnose specific issues, such as poor apex sampling of elution peaks or ion accumulation times, enabling rational and targeted optimization of the method. For instance, using DO-MS to improve apex sampling has been shown to increase ion delivery efficiency for MS2 analysis by 370% [68].
Auto-Tuning Algorithms: Recent research has focused on fully automated tuning using improved heuristic optimization algorithms, such as a Particle Swarm Optimization (PSO) algorithm enhanced with simulated annealing and dynamic boundaries [69]. This approach can automatically tune the mass spectrometer from a non-optimal state to an optimal one by finding the best balance between conflicting objectives like resolution and spectral peak intensity [69].
Diagram 2: Design of Experiments (DoE) Optimization Process
The Scientist's Toolkit: Essential Research Reagents and Materials
The following table lists key reagents and materials essential for developing and executing optimized LC-QqQ-MS methods for biomarker validation.
Table 3: Essential Research Reagent Solutions and Materials
| Item | Function/Application | Example & Notes |
|---|---|---|
| Analyte Standards | Method development and calibration | Certified reference materials for target biomarkers [67]. |
| Stable Isotope-Labeled Internal Standards (SIL-IS) | Normalize for variability in sample prep and ionization; enable precise quantification [67]. | Synthesized with ¹³C, ¹⁵N labels. |
| Mobile Phase Additives | Promote ionization and control pH; must be volatile for MS compatibility [58] [66]. | Formic Acid, Acetic Acid, Ammonium Formate, Ammonium Acetate. |
| Chromatography Columns | Separate analytes from matrix interferences to reduce ion suppression [58]. | e.g., C18, 1.7-2µm particle size, 50-150mm length. |
| Sample Preparation Kits | Clean up complex biological samples (plasma, serum) and remove interfering salts and proteins [58]. | Solid-Phase Extraction (SPE), Protein Precipitation plates. |
| Tuning and Calibration Solutions | Calibrate mass axis and optimize instrument parameters [65] [69]. | Manufacturer-provided mixes (e.g., polytyrosine). |
The meticulous optimization of collision energies and ion source parameters is a non-negotiable prerequisite for generating high-quality, reproducible data in biomarker validation research using triple quadrupole mass spectrometry. While foundational step-by-step tuning protocols remain vital, the adoption of advanced strategies such as Design of Experiments, prediction algorithms, and data-driven optimization platforms represents the current best practice. These approaches enable researchers to efficiently develop highly sensitive and robust MRM assays, even for large panels of biomarkers, thereby accelerating the pipeline from biomarker discovery to clinical validation and ultimately contributing to the advancement of personalized medicine.
Improving Signal-to-Noise Ratios through Transition Selection and Chromatography
Achieving optimal signal-to-noise (S/N) ratios is a critical determinant for the success of sensitive and reproducible assays in biomarker validation using triple quadrupole mass spectrometry. This application note details a structured framework for enhancing S/N by strategically optimizing two fundamental pillars: mass spectrometric transition selection and liquid chromatography (LC) performance. Within the context of a biomarker validation pipeline, poor S/N can jeopardize data quality, leading to inaccurate quantification and failed method acceptance criteria. We provide explicit protocols and data-driven recommendations for researchers and drug development professionals to systematically improve assay sensitivity, ensuring reliable quantification of low-abundance protein biomarkers in complex matrices.
The journey of a protein biomarker from discovery to clinical validation is a long and uncertain path [10]. The biomarker pipeline encompasses discovery, verification, and validation phases, each with distinct requirements for mass spectrometric analysis [20]. Triple quadrupole mass spectrometers, operated in Multiple Reaction Monitoring (MRM) mode, have become the benchmark for biomarker verification due to their exceptional sensitivity, specificity, and quantitative precision [10]. The core of MRM quantification lies in selecting optimal mass transitions and coupling them with high-resolution chromatography to separate the target analyte from matrix interferences.
The signal-to-noise ratio is a direct measure of an method's ability to distinguish the analyte's signal from the baseline noise, fundamentally impacting the limits of detection (LOD) and quantitation (LOQ) [70]. In regulated environments, guidelines like USP <621> provide standards for S/N calculation, though their implementation can present practical challenges for global laboratories [70]. A low S/N ratio not only raises the practical LOQ but also increases the coefficient of variation for measurements, threatening the validity of the entire biomarker study. This note addresses these challenges by providing a holistic optimization strategy focused on the two most impactful areas: mass spectrometric parameters and chromatographic separation.
Mass Spectrometric Transition Selection for Optimal Signal
In MRM assays, a "transition" refers to the specific pair of a precursor ion (Q1) and a product ion (Q3). The careful selection and optimization of these transitions is the first step toward maximizing signal and specificity.
Principles of Optimal Transition Selection
The primary goal is to select transitions that are both intense and unique to the target peptide. This process begins with the choice of a "proteotypic" peptide—a peptide that reliably represents the parent protein and is amenable to MS analysis [10]. The following criteria should guide transition selection:
- High Intensity: Choose precursor ions with the highest stable signal in Q1 MS scan.
- Unique Sequence: Ensure the peptide sequence is unique to the target protein to avoid cross-talk.
- Stable Fragmentation: Select product ions that are y-ions (typically more stable than b-ions) and appear in the higher m/z region to reduce chemical noise.
- Freedom from Interferences: Confirm the selected transition is free from isobaric interferences in the sample matrix.
Ion suppression, where the ionization of an analyte is inhibited by co-eluting compounds, is a major cause of poor signal [71]. Robust chromatography is key to mitigating this effect.
Protocol: Experimental Procedure for Transition Optimization
Purpose: To empirically identify the most intense and specific MRM transitions for a target peptide.
Materials:
- Synthetic stable isotope-labeled (SIS) peptide standard
- Triple quadrupole mass spectrometer
- Nano-flow or conventional HPLC system
- Solvent A: 0.1% Formic acid in water
- Solvent B: 0.1% Formic acid in acetonitrile
Procedure:
- Direct Infusion Tuning:
- Prepare a solution of the SIS peptide (e.g., 100 fmol/µL) in a mixture of 30% Solvent B.
- Directly infuse the solution into the mass spectrometer using a syringe pump at a flow rate of 5-10 µL/min.
- In Q1 MS mode, identify the top 2-3 most intense precursor ion charges states (typically +2 or +3).
Product Ion Scanning:
- For each candidate precursor ion, perform a product ion scan.
- Optimize collision energy (CE) by ramping voltages to determine the energy that yields the most intense fragment ions. Many software packages can automate this with scheduled CE ramps.
Transition Selection and Validation:
- From the product ion spectrum, select the 3-5 most intense fragment ions.
- Program these precursor-product ion pairs into an MRM method.
- Inject the peptide standard via LC-MRM to confirm performance under chromatographic conditions.
- Finally, inject a matrix sample (e.g., plasma) spiked with the peptide to check for specificity and select the final set of 2-3 transitions per peptide (one quantifier, others qualifiers).
Research Reagent Solutions for MS Optimization
Table 1: Key Reagents and Materials for Transition Selection and MS Analysis
| Item | Function in Experiment |
|---|---|
| Synthetic Stable Isotope-Labeled (SIS) Peptides | Serve as internal standards for both retention time alignment and precise quantification, correcting for variability in sample preparation and ionization. |
| Trypsin (Sequencing Grade) | Proteolytic enzyme used to digest proteins into peptides for bottom-up proteomics. Its high specificity and purity ensure reproducible digestion. |
| Stable Isotope Labeled Standards | |
| Formic Acid (Optima LC/MS Grade) | Mobile phase additive that promotes efficient ionization (protonation) of peptides in positive electrospray ionization mode. |
| Acetonitrile (Optima LC/MS Grade) | Organic solvent for the mobile phase; its high elution strength and volatility are ideal for LC-MS. |
Chromatographic Optimization for Noise Reduction
Chromatography is paramount for separating the analyte of interest from the complex biological matrix, thereby reducing background noise and mitigating ion suppression/enhancement effects [71]. A well-separated, sharp peak will have a higher height and a better S/N than a broad, tailing peak of the same area.
Core Strategies for Chromatographic Performance
- Solvent Mixing and Gradient Fidelity: In gradient elution, incomplete mixing of solvents can cause baseline noise and artifacts. The use of static mixers ensures uniform solvent blending before the mobile phase reaches the column, producing a stable baseline and consistent retention times [72].
- Column Chemistry and Dimensions: Shorter columns (e.g., 10-15 cm) with narrow internal diameters (e.g., 2.1 mm) and smaller particle sizes (e.g., 1.7-1.8 µm) provide higher peak efficiencies, leading to sharper peaks and improved S/N [73]. Choose the least polar column chemistry that provides adequate separation to minimize column bleed.
- Mobile Phase and Gradient Optimization: A carefully designed gradient is crucial. Ballistic (short and fast) gradients can produce sharper peaks but must be reproducible [73]. The initial oven temperature should be set about 20°C below the solvent's boiling point for effective solvent focusing at the column head [73].
Protocol: LC Method Development for Biomarker Peptides
Purpose: To develop a robust, high-resolution nano-LC or HPLC method that maximizes peak capacity and minimizes baseline noise for MRM assays.
Materials:
- C18 reversed-phase column (e.g., 15 cm x 75 µm, 1.7 µm particle size for nano-flow; 10 cm x 2.1 mm, 1.8 µm for analytical flow)
- HPLC system with binary or quaternary pumps and a well-functioning static mixer [72]
- Solvent A: 0.1% Formic acid in water
- Solvent B: 0.1% Formic acid in acetonitrile
Procedure:
- Initial Scouting Gradient:
- Start with a broad gradient from 2% to 40% Solvent B over 60 minutes at a flow rate of 300 nL/min (nano) or 0.3 mL/min (analytical).
- Inject a test digest or a cocktail of standard peptides to assess peak distribution.
Optimizing Gradient Steepness:
- If peptides are eluting too early or too late, adjust the gradient slope. For early eluters, use a shallower starting gradient. For late eluters, increase the %B at the end of the gradient.
- The goal is to achieve a relatively even distribution of peaks across the chromatographic run.
Improving Peak Shape:
- If peak tailing is observed, consider using an alternative mobile phase additive (e.g., 0.1% acetic acid) or a different column chemistry (e.g., C8).
- Ensure the column temperature is stable (e.g., 40-50°C) to improve retention time reproducibility and peak shape.
Assessing and Mitigating Noise:
- Run a blank injection (matrix without analyte) to identify regions of high background noise.
- If baseline noise is high, particularly during the gradient, verify the performance of pump seals and check for adequate degassing of solvents. The use of an efficient static mixer can significantly reduce noise related to solvent blending [72].
Workflow Diagram for S/N Optimization
The following diagram outlines the systematic workflow for improving S/N ratios, integrating both transition selection and chromatographic optimization.
Integrated Method Performance and Data Analysis
After independent optimization of MS and LC parameters, the final method must be evaluated as an integrated system. Key performance metrics should be tracked and documented.
Quantitative Data for Method Benchmarking
Table 2: Key Performance Metrics for Biomarker MRM Assay Validation
| Parameter | Target Value | Measurement Protocol |
|---|---|---|
| Signal-to-Noise Ratio (S/N) | ≥ 10 for LOQ [70] | Calculate as (2 × Signal Height) / Peak-to-Peak Noise in a blank sample near the analyte's retention time [70]. |
| Retention Time | Coefficient of Variation (CV) < 0.5% | Measure retention time across ≥ 3 injections. |
| Peak Width | Consistent; 10-30 seconds at base | Measure at 5% of peak height. |
| Peak Area Precision | CV < 15-20% (at LOQ) | Measure peak area across ≥ 3 injections of a low-level QC sample. |
Optimizing signal-to-noise ratios is not a single-step activity but a systematic process of refining both mass spectrometric and chromatographic components. For scientists engaged in the critical work of biomarker validation, a rigorous approach to transition selection and chromatographic performance is non-negotiable. By adhering to the detailed protocols for transition optimization, incorporating hardware solutions like static mixers to reduce baseline noise, and consistently monitoring the performance metrics outlined herein, researchers can significantly enhance the sensitivity and robustness of their triple quadrupole MS assays. This disciplined approach ensures that the resulting data is of sufficient quality to confidently drive decisions in the drug development pipeline.
Ensuring Long-Term Robustness and Reproducibility
The transition of protein biomarkers from discovery to clinical application requires analytical methods that are not only sensitive and specific but also robust and reproducible over the long term. Liquid chromatography coupled to triple quadrupole mass spectrometry (LC-QqQ-MS) operating in Multiple Reaction Monitoring (MRM) mode has emerged as the cornerstone technology for biomarker verification and validation due to its exceptional quantitative capabilities [3] [64]. The triple quadrupole mass analyzer is a time-honored system in biomedical research, and its use in clinical applications has increased 2-3 times in the past decade, underscoring its critical role in the field [3]. This protocol outlines comprehensive strategies and detailed methodologies to ensure the long-term robustness and reproducibility of LC-MRM-MS assays, framed within the rigorous context of biomarker validation for clinical applications.
Fundamentals of Triple Quadrupole MS for Biomarker Analysis
Triple quadrupole mass spectrometers are characterized by their tandem-in-space configuration, consisting of three quadrupoles in series (Q1-Q2-Q3) [64]. The power of this platform for quantitative analysis lies in its two stages of mass filtering. In an MRM experiment, the first quadrupole (Q1) is set to filter a specific precursor ion (typically the intact peptide ion) of a target biomarker. This selected ion is then transmitted to the second quadrupole (Q2), which acts as a collision cell. Here, the precursor ions are fragmented via collision-induced dissociation (CID) using an inert gas like argon or nitrogen [74] [64]. The resulting product ions are then passed to the third quadrupole (Q3), which is set to filter one or more specific fragment ions unique to the target peptide.
This dual mass-filtering process, monitoring a specific precursor ion → product ion transition for each analyte, confers superior specificity by effectively eliminating chemical background and interferences that would co-elute with the analyte [3] [64]. The result is a significant enhancement in signal-to-noise ratio, enabling highly sensitive and reliable quantification of biomarkers even in complex matrices like blood plasma [74]. The high specificity and sensitivity of QqQ-based MRM assays are the primary reasons it is considered the "gold standard" for targeted quantitative analysis and is extensively used in fields such as endocrine testing and newborn screening [3].
Diagram 1: MRM scanning principle in a triple quadrupole mass spectrometer.
Critical Phases for Ensuring Robustness and Reproducibility
Pre-Analytical and Experimental Design
A successful and reproducible biomarker validation study begins long before the first sample is injected into the mass spectrometer. Critical steps in experimental design are often underappreciated but are fundamental to generating meaningful and translatable data [20].
- Cohort Selection and Statistical Power: The selection of well-characterized patient and control cohorts is paramount. Cohorts must be carefully matched for potential confounders such as age, sex, and body mass index to minimize bias [20]. Furthermore, a statistical power analysis must be conducted a priori to determine the sample size required to detect a biologically significant change in biomarker levels with confidence, thereby ensuring the study is adequately powered [20].
- Sample Blinding and Randomization: To prevent analytical bias, all samples should be blinded and randomized prior to sample preparation and LC-MS analysis. This prevents the instrument operator from knowing the group identity (e.g., case vs. control) of any sample during data acquisition [20].
- Quality Control (QC) Samples: Three types of QC samples are essential for monitoring system performance and correcting for analytical drift:
- Blank QC: A sample without analyte to monitor carryover.
- Pooled QC: A representative pool of all study samples, injected at regular intervals (e.g., every 5-10 study samples) throughout the analytical batch to monitor reproducibility.
- Reference QC: A sample with a known, predetermined concentration of the target biomarkers, used for accuracy assessment and system suitability testing.
Sample Preparation Protocol
Standardized sample preparation is critical for minimizing variability. The following protocol is optimized for the depletion and processing of human blood plasma or serum, the most common biofluid for biomarker studies [75] [20].
Title: Protocol for Plasma/Serum Sample Preparation for LC-MRM-MS Biomarker Assays
Principle: This protocol describes the steps for high-abundance protein depletion, followed by protein digestion, to generate peptides suitable for LC-MRM-MS analysis, ensuring minimal technical variation.
Table 1: Research Reagent Solutions for Sample Preparation
| Reagent/Material | Function | Specification & Considerations |
|---|---|---|
| Immunoaffinity Depletion Column | Removes top 7-14 high-abundance proteins (e.g., albumin, IgG) to enhance detection of low-abundance biomarkers [75]. | Commercially available columns (e.g., MARS-14). Consider cost and sample volume requirements. |
| Magnetic Bead Kits (e.g., SP3) | For protein cleanup, digestion, and peptide purification. Efficiently handles low volumes and is amenable to automation [75]. | Kits available from PreOmics (ENRICHplus), Thermo Fisher, etc. |
| Denaturing Buffer | Unfolds proteins for efficient enzymatic digestion. | 2 M Urea or 5% SDS in Tris buffer. |
| Reducing Agent | Breaks disulfide bonds within and between proteins. | 10 mM Dithiothreitol (DTT). |
| Alkylating Agent | Caps free sulfhydryl groups to prevent reformation of disulfide bonds. | 40 mM Iodoacetamide (IAA). |
| Protease (Trypsin) | Enzymatically cleaves proteins into peptides at specific residues (Lys and Arg). | Sequencing-grade modified trypsin is recommended to minimize autolysis. |
| Solid-Phase Extraction (SPE) Plate | Desalting and cleanup of peptides post-digestion. | C18 stationary phase in 96-well format for high-throughput. |
Procedure:
- Plasma Depletion: Thaw plasma samples on ice and centrifuge at 14,000 x g for 10 minutes to remove particulates. Deplete high-abundance proteins using a commercial immunoaffinity depletion column according to the manufacturer's instructions [75].
- Protein Denaturation and Digestion: Dilute the depleted plasma 1:1 with a denaturing buffer (e.g., 2 M Urea, 50 mM Tris, pH 8.0). Reduce proteins with 10 mM DTT for 30 minutes at 37°C, then alkylate with 40 mM IAA for 30 minutes at room temperature in the dark.
- Proteolytic Digestion: Add sequencing-grade trypsin at a 1:50 (enzyme-to-protein) ratio and incubate for 12-16 hours at 37°C.
- Peptide Cleanup: Acidify the digest with 1% formic acid to stop the reaction. Purify the peptides using a C18 solid-phase extraction plate. Elute peptides with 50% acetonitrile/0.1% formic acid.
- Sample Reconstitution: Dry the purified peptides under vacuum and reconstitute them in a fixed volume of 0.1% formic acid in water. Vortex thoroughly and centrifuge before LC-MS analysis.
LC-MRM-MS Analysis and Data Acquisition
The analytical phase requires meticulous method development and consistent system monitoring.
Title: Method for LC-MRM-MS Data Acquisition for Biomarker Quantification
Instrumentation: Liquid chromatography system coupled to a triple quadrupole mass spectrometer.
Chromatography:
- Column: Reversed-phase C18 column (e.g., 2.1 x 100 mm, 1.9 µm particle size).
- Mobile Phase: A: 0.1% Formic Acid in Water; B: 0.1% Formic Acid in Acetonitrile.
- Gradient: Develop a linear gradient from 2% B to 35% B over 30-60 minutes, optimized for peptide separation.
- Column Temperature: Maintain at 40°C.
- Flow Rate: 0.2-0.3 mL/min.
Mass Spectrometry (MRM Mode):
- Transition Selection: For each target biomarker peptide, select a minimum of three MRM transitions. The most intense transition is used for quantification, while the others serve as qualifiers for confirmatory identity [64]. Transitions should be selected to avoid known interferences.
- Optimization: Use synthetic heavy isotope-labeled versions of the target peptides as internal standards to empirically optimize compound-dependent parameters like collision energy (CE) for each transition [64].
- Scheduling: Implement scheduled MRM (sMRM) to monitor each transition within a specific retention time window, thereby increasing the number of data points across a chromatographic peak and improving quantitative accuracy.
Diagram 2: End-to-end biomarker validation workflow.
Performance Monitoring and Data Analysis
Long-term reproducibility is ensured by continuous monitoring of system performance and rigorous data analysis protocols.
Table 2: Key Performance Metrics for LC-MRM-MS Assays
| Metric | Target | Purpose & Rationale |
|---|---|---|
| Retention Time Shift | < ±0.2 min | Monitors chromatographic consistency. Drift indicates column degradation or mobile phase issues. |
| Peak Area RSD (in Pooled QC) | < 15-20% | Measures precision of the analytical response. High RSD indicates instability in sample prep or instrument response. |
| Signal-to-Noise (S/N) Ratio | > 10 | Ensures detection sensitivity is maintained. A declining S/N suggests loss of sensitivity. |
| Calibration Curve R² | > 0.99 | Assesses the linearity and reliability of the quantitative response. |
| QC Accuracy | 85-115% | Verifies that the measured concentration of the reference QC is within the acceptable range of its known value. |
Data Processing and Normalization:
- Peak Integration: Integrate all MRM peaks using consistent settings across the entire batch.
- Normalization: Normalize the peak areas of the target (light) peptides to the peak areas of their corresponding stable isotope-labeled (heavy) internal standards. This corrects for variations in sample preparation and MS ionization efficiency [64].
- Batch Correction: Use the data from the pooled QC samples, injected at regular intervals, to model and correct for any systematic drift in analyte response over the acquisition batch using statistical algorithms.
Achieving long-term robustness and reproducibility in biomarker validation is an integrated process that spans from initial study design to final data analysis. By adhering to standardized protocols for sample preparation, leveraging the superior specificity of LC-MRM-MS on triple quadrupole platforms, and implementing rigorous quality control measures, researchers can generate high-quality, reliable data. This disciplined approach is essential for translating promising biomarker candidates into clinically validated tools for disease diagnosis, prognosis, and monitoring.
Ensuring Analytical Rigor: Validation Frameworks and Platform Comparisons
In the rigorous pipeline of biomarker validation, the establishment of well-defined figures of merit is a critical prerequisite for ensuring that analytical methods generate reliable, reproducible, and clinically meaningful data. For research utilizing triple quadrupole mass spectrometry (TQ-MS), typically operated in Multiple Reaction Monitoring (MRM) mode, the determination of Limit of Detection (LOD), Limit of Quantitation (LOQ), precision, and accuracy forms the foundation of analytical validation [76]. These parameters objectively characterize the sensitivity, robustness, and reliability of an assay, providing confidence in the quantitative data used to support critical decisions in drug development and clinical research. This protocol details the established methodologies for determining these essential figures of merit within the context of TQ-MS-based biomarker verification and validation.
Definitions and Theoretical Foundation
A clear understanding of the terminology is essential for proper implementation. The following definitions are aligned with guidelines from the Clinical and Laboratory Standards Institute (CLSI) and the International Council for Harmonisation (ICH) [77] [78].
- Limit of Blank (LoB): The highest apparent analyte concentration observed when replicates of a blank sample (containing no analyte) are tested. It is defined as
LoB = mean_blank + 1.645(SD_blank), assuming a Gaussian distribution where 5% of blank measurements may falsely appear positive (Type I error) [77]. - Limit of Detection (LOD): The lowest analyte concentration that can be reliably distinguished from the LoB. The LOD is not only greater than the LoB but also accounts for the variability of a low-concentration sample. It is calculated as
LOD = LoB + 1.645(SD_low concentration sample), ensuring that 95% of true low-concentration samples are detectable above the LoB [77] [78]. - Limit of Quantitation (LOQ): The lowest analyte concentration that can be quantified with acceptable precision and accuracy [77] [79]. It is the concentration at which predefined goals for bias and imprecision are met, often set at a precision of 20% coefficient of variation (CV) and accuracy of 80-120% for bioanalytical methods [79]. The LOQ cannot be lower than the LOD and is often significantly higher.
- Precision: The closeness of agreement between a series of measurements obtained from multiple sampling of the same homogeneous sample under prescribed conditions. It is typically expressed as the coefficient of variation (%CV) of repeated measurements [79] [78].
- Accuracy: The closeness of agreement between the measured value of an analyte and its true value. It is typically expressed as the percentage difference (bias) between the measured mean and the accepted true value [79].
The conceptual relationship and typical distribution of results for these key figures of merit are illustrated in the following workflow.
Experimental Protocols for Determining Figures of Merit
The following section provides detailed methodologies for the experimental determination of LOD, LOQ, precision, and accuracy. A critical first step is the preparation of matrix-matched quality control (QC) samples, as the use of a authentic biological matrix is essential for determining method LOD/LOQ, rather than an instrumental LOD/LOQ, which can be overly optimistic [78].
Sample Preparation and Calibration Standards
- Matrix Selection: Use the same biological matrix as the intended study samples (e.g., human plasma, serum, urine). Ensure the blank matrix is confirmed to be free of the target analyte(s) [80] [76].
- QC Sample Preparation: Spike a known amount of the analyte and stable isotope-labeled internal standard (SIL-IS) into the blank matrix. For LOD/LOQ determinations, prepare at least 20 replicates of a low-concentration QC sample. For precision and accuracy, prepare QC samples at three concentrations (low, mid, and high) across the calibration curve range, with a minimum of 5 replicates per level [77] [79].
- Calibration Curve: Prepare a calibration curve with a minimum of six non-zero concentrations, analyzed in duplicate or triplicate. The curve should cover the entire expected concentration range, from the lower LOQ (LLOQ) to the upper LOQ (ULOQ) [79].
Protocol for Limit of Detection (LOD) and Limit of Blank (LoB)
This protocol follows the CLSI EP17 recommended approach [77].
Analyze Blank and Low-Concentration Samples:
- Analyze a minimum of 20 independent replicates of the blank sample (containing no analyte) and 20 replicates of a sample containing a low concentration of analyte (estimated to be near the LOD).
- Process all samples through the complete analytical procedure, including sample preparation.
Calculate LoB:
- Calculate the mean and standard deviation (SD) of the results from the blank samples.
LoB = mean_blank + 1.645 * SD_blank(for a one-sided 95% confidence level).
Calculate LOD:
- Calculate the mean and SD of the results from the low-concentration sample.
LOD = LoB + 1.645 * SD_low concentration sample.
Verification: Confirm the calculated LOD by analyzing a sufficient number of samples (e.g., n=20) spiked at the LOD concentration. The LOD is verified if no more than 5% of the results fall below the LoB [77].
Protocol for Limit of Quantitation (LOQ)
The LOQ can be determined using several approaches. The following table summarizes the most common methods.
Table 1: Common Approaches for Determining the Limit of Quantitation (LOQ)
| Approach | Description | Calculation / Criteria | Applicability |
|---|---|---|---|
| Precision & Accuracy-Based [79] | Based on the concentration that meets predefined precision and accuracy goals. | Analyze replicates (n≥5) at low concentrations. LOQ is the lowest concentration where CV ≤ 20% and accuracy (bias) within ±20%. | Universal; recommended by most bioanalytical guidelines. |
| Signal-to-Noise (S/N) Ratio [79] | The analyte concentration that produces a signal distinguishable from the background noise. | S/N ratio ≥ 10:1. Noise is measured as the variability of the baseline near the analyte's retention time. | Chromatographic methods (LC-MS, HPLC). Simple but can be instrument-dependent. |
| Calibration Curve [79] | Uses the standard error of the calibration curve and its slope. | LOQ = 10 * (SD_response / Slope). SD can be the standard deviation of the blank, the y-intercept, or the regression. |
Requires a linear calibration curve with concentrations near the expected LOQ. |
Protocol for Precision and Accuracy
- Sample Analysis: Analyze the QC samples (low, mid, high) in at least 5 replicates per run. Repeat the analysis over at least three separate days to capture inter-day (between-day) variability [80].
- Calculate Precision: Precision is expressed as the Coefficient of Variation (%CV).
- Within-Day Precision: Calculate the mean and SD for the replicates at each QC level within a single analytical run.
%CV = (SD / Mean) * 100. - Between-Day Precision: Calculate the mean and SD using the data from all replicates across all analytical runs for each QC level.
- Within-Day Precision: Calculate the mean and SD for the replicates at each QC level within a single analytical run.
- Calculate Accuracy: Accuracy is expressed as the percentage difference from the nominal (theoretical) concentration.
Accuracy (%) = (Mean measured concentration / Nominal concentration) * 100.- Bias (%) = 100 - Accuracy (%)`.
- Acceptance Criteria: For bioanalytical methods, typical acceptance criteria are:
Workflow Integration in Biomarker Validation
The determination of figures of merit is not an isolated activity but is integrated into the broader biomarker validation pipeline, which spans from discovery to clinical application. The role of TQ-MS and MRM assays is particularly critical in the verification phase, which serves as a bridge between discovery and large-scale clinical validation [10] [76]. The following diagram illustrates this workflow and the placement of analytical validation.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table details key reagents and materials essential for successfully establishing figures of merit in TQ-MS biomarker assays.
Table 2: Essential Research Reagents and Materials for TQ-MS Biomarker Assay Validation
| Item | Function and Importance | Considerations for Validation |
|---|---|---|
| Stable Isotope-Labeled Internal Standards (SIL-IS) [76] | Corrects for sample preparation losses, matrix effects, and instrument variability, critically improving precision and accuracy. | Ideally, the SIL-IS should be a peptide/protein for protein biomarkers. Use a concentration near the mid-point of the calibration curve. |
| Authentic Biological Matrix [78] | Serves as the sample diluent for calibration standards and QCs. Essential for determining the method LOD/LOQ, which includes matrix effects. | Source should be consistent and well-characterized. Use pooled matrix from multiple donors to minimize individual variability. |
| Synthetic/Authentic Analyte Standard | Used to prepare calibration curves and QC samples for accurate quantification. | Purity must be well-characterized and certified. Used to spike into the blank matrix. |
| Pass-Through Phospholipid Removal Plates (e.g., Ostro) [80] | Advanced sample clean-up that combines protein precipitation with efficient removal of phospholipids, reducing matrix effects and improving assay sensitivity and reproducibility. | Superior to traditional protein precipitation alone, leading to more consistent recovery and lower background noise. |
| Quality Control (QC) Materials | Independently prepared samples used to monitor the performance of the analytical run. | Should be prepared in the same matrix as study samples, at low, mid, and high concentrations. Used to accept or reject an analytical run. |
Data Presentation and Analysis
Summarizing the results of the validation is crucial for reporting and regulatory submissions. The following template can be used to present the key figures of merit for a representative analyte.
Table 3: Representative Validation Data for a Hypothetical Protein Biomarker Analyte
| Validation Parameter | QC Level (Low / Mid / High) | Result | Acceptance Criteria Met? |
|---|---|---|---|
| Calibration Curve Range | N/A | 1 - 500 ng/mL | N/A |
| Lower LOQ (LLOQ) | 1 ng/mL | 1 ng/mL | Yes |
| Precision (Within-Day, %CV, n=5) | Low (1 ng/mL) | 5.2% | ≤ 20% ✓ |
| Mid (50 ng/mL) | 4.1% | ≤ 15% ✓ | |
| High (400 ng/mL) | 3.8% | ≤ 15% ✓ | |
| Precision (Between-Day, %CV, n=15) | Low (1 ng/mL) | 7.5% | ≤ 20% ✓ |
| Mid (50 ng/mL) | 6.2% | ≤ 15% ✓ | |
| High (400 ng/mL) | 5.9% | ≤ 15% ✓ | |
| Accuracy (Within-Day, % Bias, n=5) | Low (1 ng/mL) | +4.5% | ± 20% ✓ |
| Mid (50 ng/mL) | -2.1% | ± 15% ✓ | |
| High (400 ng/mL) | +3.8% | ± 15% ✓ | |
| Accuracy (Between-Day, % Bias, n=15) | Low (1 ng/mL) | +5.5% | ± 20% ✓ |
| Mid (50 ng/mL) | -3.2% | ± 15% ✓ | |
| High (400 ng/mL) | +4.1% | ± 15% ✓ | |
| Limit of Detection (LOD) | N/A | 0.25 ng/mL | N/A |
Within the critical pathway of biomarker validation and drug development, the demand for analytical techniques that offer both high specificity and reliable quantification is paramount. For decades, immunoassays, such as the enzyme-linked immunosorbent assay (ELISA), have been the cornerstone of protein quantification in biological samples due to their high sensitivity and throughput [81] [29]. However, these antibody-based methods possess inherent limitations, including the potential for antibody cross-reactivity and the significant time and cost required to develop new assays [4] [82] [29]. In contrast, triple quadrupole mass spectrometry coupled with selected reaction monitoring (QqQ-SRM) has emerged as a powerful alternative, offering superior specificity, the ability to multiplex, and a faster transition from assay development to deployment [4] [82]. This application note provides a detailed benchmark of QqQ-SRM against traditional immunoassays, underscoring its advantages in the context of biomarker verification and clinical research, and includes a comprehensive protocol for implementing a QqQ-SRM assay.
Comparative Performance: QqQ-SRM vs. Immunoassays
The selection of an analytical technique is a critical decision in experimental design. The table below summarizes the core characteristics of QqQ-SRM and immunoassays, highlighting the distinct advantages of the mass spectrometry-based approach for targeted protein quantification.
Table 1: Key Characteristics of QqQ-SRM and Immunoassays
| Characteristic | QqQ-SRM/MRM | Immunoassays (e.g., ELISA) |
|---|---|---|
| Analytical Principle | Physical separation by mass-to-charge ratio (m/z) of proteotypic peptides and their fragments [81] [82]. | Molecular recognition using antibody-antigen binding [29]. |
| Specificity | Very high; capable of distinguishing protein isoforms, post-translational modifications, and genetic variants [83] [82]. | Variable; susceptible to cross-reactivity with homologous proteins or matrix components [4] [29]. |
| Multiplexing Capacity | High; can routinely quantify >100 proteins in a single LC-MS run [81] [83]. | Low to moderate; traditional ELISA is single-plex. Newer platforms (e.g., Luminex) allow multiplexing but with challenges in antibody compatibility [29]. |
| Assay Development | Relatively rapid and cost-effective; does not require specific antibodies, relies on synthetic peptides [82]. | Time-consuming (can take a year) and expensive ($50,000-$100,000 per assay); requires production and validation of high-quality antibodies [81] [29]. |
| Throughput | High for multiplexed analysis; lower for single proteins without automation. | Very high for single-plex analysis of many samples [29]. |
| Dynamic Range | Typically 3-4 orders of magnitude [82]. | Wide, up to 4-5 orders of magnitude for some platforms like MSD [29]. |
| Sensitivity | Can reach low ng/mL to sub-ng/mL in plasma with enrichment; generally less sensitive than the best immunoassays for single-plex analysis [81] [29]. | Excellent; can detect proteins in the pg/mL range [81] [29]. |
A key demonstration of specificity comes from direct comparative studies. For instance, an analysis of 11 steroids in human plasma showed that while both QqQ-SRM and time-of-flight (TOF)-MS could detect the analytes, the primary challenge for the TOF platform was selectivity rather than sensitivity, underscoring the robust performance of the targeted QqQ approach in complex matrices [84]. Furthermore, the shift in fields like endocrine testing towards LC-SRM is driven by its superior specificity and accuracy compared to immunoassays, which can yield overestimated concentrations due to antibody cross-reactivity [4].
QqQ-SRM Experimental Workflow
A robust QqQ-SRM assay involves a series of deliberate steps, from sample preparation to data analysis. The following diagram and protocol outline the entire process.
Figure 1: QqQ-SRM Targeted Proteomics Workflow. The process involves sample preparation, LC separation, and highly specific mass analysis using a triple quadrupole mass spectrometer.
Step-by-Step Protocol
Protocol: Development and Execution of a QqQ-SRM Assay for Targeted Protein Quantification
I. Sample Preparation (Critical for Sensitivity and Reproducibility)
- Protein Extraction and Denaturation: Extract proteins from your biological matrix (e.g., plasma, tissue, cells). Use a buffer containing 8 M urea or 5% SDS to denature proteins and disrupt complex structures.
- Reduction and Alkylation: Reduce disulfide bonds with 10 mM dithiothreitol (DTT) at 37°C for 45 minutes. Alkylate free thiols with 25 mM iodoacetamide (IAA) at room temperature in the dark for 30 minutes.
- Protein Digestion: Dilute the sample to reduce denaturant concentration. Add sequencing-grade trypsin at a 1:25-1:50 (enzyme-to-protein) ratio and incubate at 37°C overnight [85]. Acidify the digest with 1% trifluoroacetic acid (TFA) to stop the reaction.
- Desalting: Purify the resulting peptides using C18 solid-phase extraction (SPE) cartridges. Elute peptides in a solution of 50-80% acetonitrile with 0.1% formic acid, and dry down using a vacuum concentrator.
II. Assay Configuration and LC-SRM Setup
- Selection of Proteotypic Peptides: For each target protein, select 2-3 unique "proteotypic" peptides that are specific to the protein and exhibit good MS detectability. Databases such as SRMAtlas or PeptideAtlas can be consulted [82]. Avoid peptides with variable modifications or missed cleavages.
- Transition Selection: For each proteotypic peptide, select a precursor ion (typically a doubly or triply charged ion) and 3-5 characteristic fragment (product) ions. The pair of a specific precursor and product ion is a "transition." Use spectral libraries or experimental data to choose the most intense, unique fragments [82].
- Synthesis of Internal Standards: Synthesize stable isotope-labeled (SIL) versions of each proteotypic peptide (e.g., containing 13C/15N-labeled Arginine or Lysine) to be used as internal standards. These are spiked into the sample at a known concentration to account for sample loss and ionization variability [82].
- Liquid Chromatography: Reconstitute the dried peptide digest in 0.1% formic acid. Separate peptides using reversed-phase nano-liquid chromatography (nano-LC) or high-performance liquid chromatography (HPLC) with a C18 column and a gradient of increasing organic solvent (acetonitrile).
- Mass Spectrometry Data Acquisition:
- Instrument: Triple quadrupole mass spectrometer.
- Ionization: Electrospray ionization (ESI) in positive mode.
- SRM Method: Program the mass spectrometer to monitor the predefined transitions for both the native (light) and SIL (heavy) peptides within specific retention time windows. The first quadrupole (Q1) selects the precursor ion, the second (Q2) fragments it via collision-induced dissociation, and the third quadrupole (Q3) selects a specific product ion for detection [81] [82].
III. Data Analysis
- Peak Integration: Integrate the chromatographic peaks for each transition of the light and heavy peptides.
- Quantification: Calculate the peak area ratio of light to heavy for each peptide. Use a calibration curve, created by spiking known amounts of heavy peptide into a constant matrix background, to determine the absolute concentration of the target protein in the original sample [82].
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of a QqQ-SRM assay relies on a set of key reagents and materials. The following table details these essential components.
Table 2: Key Research Reagent Solutions for QqQ-SRM
| Reagent / Material | Function | Critical Considerations |
|---|---|---|
| Sequencing-Grade Trypsin | Proteolytic enzyme that digests proteins into peptides for mass analysis. | Ensures specific and complete cleavage at lysine and arginine residues, minimizing missed cleavages that complicate analysis [83]. |
| Stable Isotope-Labeled (SIL) Peptides | Internal standards for precise and accurate quantification. | Spiked into the sample pre-digestion; corrects for losses during sample preparation and variability in MS ionization efficiency. Essential for absolute quantification [82] [85]. |
| C18 Solid-Phase Extraction (SPE) Cartridges | Desalting and purification of peptide mixtures after digestion. | Removes detergents, salts, and other impurities that can suppress ionization and interfere with LC-MS analysis [85]. |
| Reversed-Phase LC Column | Chromatographic separation of peptides prior to MS injection. | Nano-flow C18 columns are standard for optimal sensitivity. The column length and particle size impact resolution and throughput [83]. |
| High-Affinity Antibody Beads (for SISCAPA) | Immunoaffinity enrichment of target peptides from complex digests. | Not always required, but can dramatically enhance sensitivity by enriching low-abundance target peptides, pushing detection limits to the pg/mL range [86]. |
The superior specificity of QqQ-SRM, combined with its multiplexing capabilities and relatively streamlined assay development, positions it as an indispensable tool for modern biomarker validation and translational research. While immunoassays remain the gold standard for high-sensitivity, high-throughput analysis of single proteins where well-validated kits exist, QqQ-SRM provides a compelling alternative for projects requiring the unambiguous quantification of multiple specific protein targets, including isoforms and post-translationally modified forms. As mass spectrometry technology continues to evolve, with the emergence of hybrid platforms like the Stellar MS that combine QqQ robustness with high-resolution scanning, the gap between discovery proteomics and routine clinical application will continue to narrow, further solidifying the role of mass spectrometry in the future of molecular diagnostics and personalized medicine [85].
Comparing QqQ with High-Resolution Platforms like Q-TOF and Orbitrap
The selection of an appropriate mass spectrometry platform is a critical decision in the design of biomarker validation pipelines. Triple quadrupole (QqQ) mass spectrometers have long been the gold standard for targeted quantification in biomarker research, while high-resolution accurate-mass (HRAM) platforms such as quadrupole time-of-flight (Q-TOF) and Orbitrap instruments offer distinct advantages for untargeted discovery and confirmation [4] [87]. This technical note provides a detailed comparison of these platforms within the context of biomarker validation, including performance characteristics, application-specific workflows, and experimental protocols to guide researchers in selecting the optimal technology for their specific research requirements.
Technical Comparison of Mass Spectrometry Platforms
Fundamental Operating Principles
Triple Quadrupole (QqQ) MS utilizes three sequential quadrupoles: Q1 for precursor ion selection, Q2 as a collision cell for fragmentation, and Q3 for product ion analysis. This configuration enables highly specific Selected Reaction Monitoring (SRM) transitions, where a specific precursor ion and one of its fragment ions are monitored, providing exceptional sensitivity and selectivity for targeted quantification [24] [4].
Q-TOF MS combines a quadrupole mass filter with a time-of-flight mass analyzer. The quadrupole (Q1) can operate in either mass selection mode or RF-only mode to transmit all ions. The TOF analyzer separates ions based on their time of flight through a field-free drift region, providing high-resolution and accurate mass capabilities for both precursor and product ions [88].
Orbitrap MS utilizes an electrostatic orbital trap where ions oscillate around a central spindle electrode. The measurement of image current from these oscillations, followed by Fourier transformation, enables very high resolution and mass accuracy [89] [90]. Commercial Orbitrap instruments are typically hybrid systems, often combined with quadrupole or linear ion trap mass analyzers for enhanced functionality [89] [91].
Performance Characteristics Comparison
Table 1: Key Performance Metrics for Different Mass Spectrometry Platforms
| Parameter | Triple Quadrupole (QqQ) | Q-TOF | Orbitrap |
|---|---|---|---|
| Mass Accuracy | Unit mass resolution | Typically <5 ppm | Sub-1 ppm with internal calibration [89] |
| Resolving Power | Unit mass resolution | Typically 20,000-80,000 | 120,000 to 1,000,000 FWHM [89] [91] |
| Quantitative Sensitivity | Excellent (fg-pg level) [87] | Good | Good to excellent |
| Dynamic Range | Up to 5 orders of magnitude [91] | 4-5 orders of magnitude | Up to 5 orders of magnitude [91] |
| Scan Speed | Moderate | Very High (up to 100 Hz) | Moderate to High (up to 40 Hz) [91] |
| Fragmentation Capabilities | CID, SRM, MRM | CID, Data-dependent MS/MS | CID, HCD, ETD, UVPD [91] [87] |
Table 2: Application Strengths and Limitations of Different MS Platforms
| Aspect | Triple Quadrupole (QqQ) | Q-TOF | Orbitrap |
|---|---|---|---|
| Key Strengths | Superior sensitivity for targeted quantification; Robust and reproducible; Lower instrument cost [4] [87] | Fast acquisition; Accurate mass; Untargeted screening capability; Rapid method updating [88] | Ultra-high resolution; Excellent mass accuracy; Wide applications from small molecules to proteins [89] |
| Key Limitations | Unit mass resolution; Limited untargeted capability | Slightly lower sensitivity vs. QqQ for targeted work; Higher cost than QqQ [92] | High instrument cost; Complex operation [87] |
| Ideal Biomarker Workflow Stage | Validation & Verification | Discovery & Preliminary Validation | Discovery & Confirmatory Analysis |
Application-Specific Considerations for Biomarker Research
Targeted Biomarker Validation
For targeted biomarker validation, where the objective is precise quantification of predefined analytes, QqQ platforms operating in SRM mode remain the preferred choice [24] [4]. The exceptional sensitivity and specificity of SRM transitions enable reliable detection and quantification of low-abundance biomarkers in complex matrices. The two stages of mass filtering in SRM significantly reduce chemical background, providing a sensitivity increase of one to two orders of magnitude compared to full scan methods [24].
In proteomic biomarker validation, SRM typically targets proteotypic peptides (PTPs) that uniquely represent the protein of interest. These peptides must exhibit good ionization efficiency, fall within the mass range of the instrument, and demonstrate predictable fragmentation to generate specific transitions for monitoring [24].
Untargeted Biomarker Discovery
For untargeted biomarker discovery, Q-TOF and Orbitrap platforms offer significant advantages due to their high-resolution and accurate-mass capabilities [89] [88]. The ability to collect full-scan HRAM data during untargeted analysis facilitates the identification of novel compounds and enables retrospective data analysis without the need to re-run samples [89].
Q-TOF instruments are particularly valuable in applications requiring rapid screening of unknown compounds, such as in toxicology and metabolomics, where the continuous emergence of new analytes presents analytical challenges [88]. The high mass accuracy and resolution of both Q-TOF and Orbitrap systems enable differentiation of isobaric compounds that would be indistinguishable using unit mass resolution instruments [89].
Hybrid Approaches for Comprehensive Biomarker Workflows
Integrated biomarker research often benefits from a complementary approach utilizing both QqQ and HRAM platforms at different stages of the workflow. A typical pipeline might employ:
- Discovery Phase: Q-TOF or Orbitrap for untargeted analysis to identify potential biomarker candidates [89] [88].
- Validation Phase: QqQ-SRM for targeted, high-throughput validation of candidate biomarkers across large sample sets [24] [4].
- Confirmatory Analysis: Orbitrap for structural elucidation and definitive identification of biomarkers [89].
This integrated approach leverages the distinct strengths of each platform throughout the biomarker development pipeline.
Experimental Protocols
Protocol 1: Biomarker Validation Using QqQ-SRM
Objective: Targeted quantification of candidate protein biomarkers in biological samples using triple quadrupole MS in SRM mode.
Materials and Reagents:
- Internal Standards: Stable isotope-labeled peptide analogs for each target proteotypic peptide.
- Digestion Enzymes: Trypsin (sequencing grade) or immobilized enzyme kits to minimize enzyme contamination [42].
- Solid-Phase Extraction: C18 cartridges or plates for sample cleanup.
- LC Mobile Phases: Solvent A: 0.1% formic acid in water; Solvent B: 0.1% formic acid in acetonitrile.
Procedure:
- Sample Preparation:
- Precipitate proteins from biofluids (e.g., urine, plasma) using appropriate methods (e.g., acetone, TCA) [42].
- Reduce with dithiothreitol and alkylate with iodoacetamide.
- Digest with trypsin (1:20-1:50 enzyme-to-protein ratio) at 37°C for 12-16 hours.
SPE Cleanup:
- Condition C18 cartridges with methanol and equilibrate with 0.1% formic acid.
- Load digested samples, wash with 0.1% formic acid, and elute with 50-80% acetonitrile containing 0.1% formic acid.
- Concentrate samples using vacuum centrifugation and reconstitute in 0.1% formic acid for LC-MS analysis.
LC-SRM/MS Analysis:
- Chromatography: Utilize reversed-phase nanoLC or UHPLC with C18 column (75μm × 15cm, 2μm particle size) with a 30-60 minute gradient from 2% to 35% solvent B.
- SRM Method Development:
- Select 2-3 proteotypic peptides per protein biomarker.
- For each peptide, identify 3-5 optimal SRM transitions using synthetic peptides.
- Prioritize y-ion fragments with m/z higher than precursor m/z to enhance specificity [24].
- Include heavy isotope-labeled internal standards for each target peptide.
- Mass Spectrometry Parameters:
- Q1 and Q3 resolution: 0.7 Da FWHM
- Dwell time: 10-50 ms per transition
- Collision energy: Optimized for each peptide
Data Analysis:
- Integrate peak areas for each transition.
- Calculate peak area ratios (light/heavy) for quantification.
- Monitor at least 3-5 data points per peak for reliable quantification.
Figure 1: QqQ-SRM Biomarker Validation Workflow
Protocol 2: Untargeted Biomarker Discovery Using Q-TOF MS
Objective: Comprehensive profiling of small molecules or peptides for biomarker discovery using Q-TOF mass spectrometry.
Materials and Reagents:
- Quality Controls: Pooled quality control samples for system suitability assessment.
- Protein Removal: Precipitation reagents (methanol, acetonitrile) or ultrafiltration devices.
- LC Columns: Reversed-phase C18 columns (2.1 × 100 mm, 1.7-1.8 μm) for small molecules; C8 or C4 for lipids.
- Reference Standards: Mass calibration standards for instrument calibration.
Procedure:
- Sample Preparation:
- For metabolomics: Precipitate proteins with cold methanol or acetonitrile (2:1-4:1 solvent-to-sample ratio).
- For peptidomics: Process samples using appropriate enrichment methods if analyzing low-abundance peptides.
- Include pooled quality control samples by combining equal aliquots from all test samples.
LC-Q-TOF/MS Analysis:
- Chromatography: Utilize UHPLC with C18 column and 10-20 minute gradient for high-throughput analysis.
- Mass Spectrometry Parameters:
- Acquisition Mode: Data-independent acquisition (DIA) such as SWATH or MSE [88].
- Mass Range: m/z 50-1200 for small molecules; m/z 300-2000 for peptides.
- Collision Energy: Low energy (4-10 eV) for precursor ions; ramped high energy (15-40 eV) for fragment ions.
- Reference Lock Mass: Use reference compound for real-time mass calibration.
Data Processing and Analysis:
- Perform peak picking, alignment, and normalization using specialized software.
- Use statistical analysis (PCA, OPLS-DA) to identify significantly altered features.
- Conduct database searching for metabolite identification using accurate mass and fragmentation patterns.
Figure 2: Q-TOF Untargeted Biomarker Discovery Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for Biomarker Mass Spectrometry
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Stable Isotope-Labeled Internal Standards | Normalization for quantification; Compensation for sample preparation variability | Essential for accurate QqQ-SRM quantification; Should be added early in sample preparation [24] |
| Immobilized Enzymes | Protein digestion without enzyme contamination; Improved digestion efficiency | Particularly valuable for low-abundance biomarkers; Reduces background interference [42] |
| Solid-Phase Extraction Cartridges | Sample cleanup; Analyte concentration; Matrix component removal | C18 for most peptides and small molecules; Alternative chemistries for specific analyte classes |
| UHPLC Columns | High-resolution chromatographic separation; Reduced analysis time | Sub-2μm particles for improved resolution; Various chemistries (C18, HILIC, etc.) for different analytes |
| Quality Control Materials | System suitability assessment; Data quality monitoring | Pooled patient samples; Commercial quality control materials; Processed sample aliquots |
The selection between QqQ, Q-TOF, and Orbitrap platforms for biomarker research depends heavily on the specific stage of the biomarker pipeline and the analytical requirements of the study. QqQ systems provide unmatched sensitivity and precision for targeted validation of known biomarkers across large sample sets, making them ideal for verification and clinical application stages. In contrast, Q-TOF and Orbitrap platforms offer superior capabilities for untargeted discovery and comprehensive characterization, with high resolution and mass accuracy enabling definitive identification of novel biomarkers.
A strategic integration of both platform types throughout the biomarker development pipeline—using HRAM instruments for discovery and preliminary identification, followed by QqQ-SRM for high-throughput validation—represents the most effective approach for robust biomarker research. This complementary methodology leverages the distinct advantages of each technology to maximize both the breadth of biomarker discovery and the rigor of validation.
- Introduction & Clinical Need: Overview of DKD diagnostic limitations and PromarkerD solution.
- Biomarker Identification & Verification: Discovery of APOA4, CD5L, IGFBP3 panel and verification methods.
- Validation Study Design: Multi-phase validation across 572 patients with standardized protocols.
- Analytical Performance: MRM assay precision data and clinical validation results.
- Clinical Utility: Predictive performance in T1D and T2D populations.
- Experimental Protocols: Detailed sample preparation, LC-MRM/MS, and data analysis methods.
- Pathway Diagrams & Reagents: Visual workflows and essential research materials.
Case Study: Successful Clinical Validation of a Protein Panel for Diabetic Kidney Disease
Diabetic kidney disease (DKD) represents one of the most prevalent and serious complications of diabetes, affecting approximately 20-30% of all diabetic patients and ranking as the leading cause of end-stage renal disease worldwide [48] [93]. The current gold-standard diagnostics for DKD - urinary albumin-to-creatinine ratio (UACR) and estimated glomerular filtration rate (eGFR) - demonstrate significant clinical limitations, including poor sensitivity for early-stage detection and inability to reliably predict disease progression [48] [94]. These diagnostic shortcomings create a critical clinical gap in effective DKD management, as early intervention can substantially slow renal function decline before irreversible damage occurs [48].
The PromarkerD biomarker test was developed to address this unmet clinical need through a robust mass spectrometry-based proteomic approach. This validated protein biomarker panel offers a novel prognostic capability - predicting the risk of incident CKD or significant renal function decline (≥30% eGFR reduction) within a four-year timeframe, enabling earlier clinical intervention and personalized patient management strategies [95].
Biomarker Identification & Verification
Biomarker Panel Discovery
The PromarkerD biomarker panel was discovered through a comprehensive proteomic analysis of plasma samples from well-characterized patients at different stages of diabetic kidney disease [48]. Using a dual-mass spectrometry approach, researchers employed iTRAQ (isobaric tags for relative and absolute quantitation) labeling for initial discovery phase analysis on pooled samples, followed by multiple reaction monitoring (MRM) for verification and validation across large patient cohorts [48] [96]. Through this rigorous process, three key protein biomarkers were identified and validated:
Table 1: PromarkerD Protein Biomarker Panel
| Protein Biomarker | Full Name | Biological Significance | Association with DKD |
|---|---|---|---|
| APOA4 | Apolipoprotein A-IV | Lipid metabolism, reverse cholesterol transport | Significantly elevated in DKD progressors [95] |
| CD5L | CD5 antigen-like | Immune regulation, apoptosis control | Significantly elevated in DKD progressors [95] |
| IGFBP3 | Insulin-like growth factor-binding protein 3 | IGF transport, cell growth regulation | Combined with other parameters for risk prediction [95] |
Verification and Assay Development
The verification phase established MRM assays for each candidate biomarker, demonstrating exceptional analytical precision with intra-day and inter-day coefficients of variation of 5.9% and 8.1% respectively [48]. The final algorithm incorporates the concentrations of all three protein biomarkers combined with clinical parameters (age, HDL-cholesterol, and eGFR) to generate a personalized risk score ranging from 0-100% [95]. This score categorizes patients into distinct risk groups: low (<10%), moderate (10% to <20%), or high (≥20%) risk of developing incident CKD or experiencing rapid renal decline within four years [95].
Validation Study Design
Cohort Design and Patient Selection
The clinical validation of PromarkerD followed a multi-phase approach with increasing cohort sizes to ensure statistical robustness and clinical relevance [48]. The study utilized samples from the Fremantle Diabetes Study (FDS), a community-based, longitudinal, observational study with comprehensive biochemical testing and sample collection [48] [95]. The validation strategy progressed through three distinct phases:
Table 2: Validation Study Cohort Design
| Study Phase | Sample Size | Patient Characteristics | Primary Objectives |
|---|---|---|---|
| Discovery | 60 patients (20 per group) | T2D patients stratified by albuminuria status | Initial biomarker identification using iTRAQ proteomics |
| Preliminary Validation | 30 patients (10 per group) | New T2D patients across albuminuria categories | MRM assay verification on independent samples |
| Analytical Validation | 572 patients | Independent T2D patients from FDS cohort | Clinical validation across large population |
Clinical Endpoints and Statistical Analysis
The primary clinical endpoints for validation included either development of incident chronic kidney disease (eGFR < 60 mL/min/1.73m² in patients without baseline CKD) or an eGFR decline of ≥30% over a four-year follow-up period [95]. Statistical performance was assessed using the area under the receiver operating characteristic curve (ROC AUC), with additional calculation of sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) at pre-specified risk cut-offs [95].
Analytical Performance & Clinical Validation
MRM Assay Performance
The targeted mass spectrometry approach using multiple reaction monitoring demonstrated exceptional analytical performance suitable for clinical application [48]. The triple-quadrupole platform enabled highly specific and sensitive quantitation of the three protein biomarkers in plasma samples, with the assay demonstrating robust precision across both intra-day (5.9% CV) and inter-day (8.1% CV) measurements [48]. This high level of analytical precision ensured reliable biomarker quantitation across the large validation cohort and supported the clinical utility of the test.
Clinical Validation Results
The clinical validation demonstrated outstanding performance in predicting renal function decline:
Table 3: Clinical Validation Performance of PromarkerD
| Performance Metric | Type 2 Diabetes Cohort | Type 1 Diabetes Cohort | Clinical Significance |
|---|---|---|---|
| ROC AUC | 0.77 (CKD Stage ≥1) [48] | 0.93 (95% CI: 0.87-0.99) [95] | Excellent predictive accuracy |
| Moderate Risk Cut-off (10% to <20%) | Sensitivity + Specificity = 168.2% [95] | PPV: 46.7%, NPV: ≥92.0% [95] | Good clinical utility |
| High Risk Cut-off (≥20%) | Not reported | PPV: 50.0%, NPV: ≥92.0% [95] | Moderate PPV, excellent NPV |
| Sample Type | Plasma [48] | Plasma [95] | Standard collection protocol |
The validation in type 1 diabetes patients demonstrated particularly impressive performance, with an AUC of 0.93, suggesting the test performs at least as well in T1D as in the original T2D population [95]. The high negative predictive value (≥92%) at both risk cut-offs is clinically significant, enabling confident identification of patients unlikely to experience renal decline who may not require intensive monitoring [95].
Clinical Utility and Implementation
Predictive Performance in Diabetic Populations
The validated PromarkerD test provides a significant advance over standard clinical parameters by offering a prognostic capability that identifies at-risk patients before significant renal decline occurs. In the type 1 diabetes cohort, the test demonstrated exceptional predictive power, with PromarkerD scores substantially higher in those who experienced adverse renal outcomes (median 12.18%) compared to those who did not (median 0.16%) [95]. This strong discriminatory capability enables clinicians to stratify patients based on individual progression risk rather than relying solely on current renal function metrics.
Advantages Over Current Standards
The key advantages of the PromarkerD biomarker panel compared to current standard tests include:
- Early prediction: Capability to identify patients at risk of renal decline up to four years before it occurs [95]
- Superior performance: Outperforms current gold standard tests (UACR and eGFR) for DKD prediction [48]
- Dual diabetes applicability: Validated in both type 1 and type 2 diabetic populations [48] [95]
- Quantitative risk stratification: Provides individualized risk scores rather than binary outcomes [95]
- Robust platform: MRM mass spectrometry offers high specificity and multiplexing capability [48]
Experimental Protocols
Sample Preparation Protocol
Plasma sample processing follows a standardized protocol to ensure reproducible biomarker quantification:
- Immunodepletion: Remove 14 most abundant plasma proteins using a Human 14 Multiple Affinity Removal System (MARS14) column (Agilent Technologies) [48]
- Protein Digestion: Digest immunodepleted proteins with trypsin following standard protocols [48]
- iTRAQ Labeling (Discovery Phase): Label tryptic peptides with iTRAQ reagents (Sciex) according to manufacturer's protocol [48]
- Peptide Cleanup: Desalt labeled peptides using Strata-X 33 μM polymeric reversed phase column (Phenomenex) [48]
LC-MRM/MS Analysis
Liquid Chromatography Conditions:
- System: Agilent 1100 HPLC system or equivalent
- Column: C18 reversed-phase column
- Gradient: 10-40% acetonitrile in 0.1% trifluoroacetic acid over 60-90 minutes
- Flow Rate: 300 nL/min for nanoflow configurations [48]
Mass Spectrometry Parameters:
- Platform: Triple quadrupole mass spectrometer
- Ionization Source: Nano-electrospray ionization
- Acquisition Mode: Multiple Reaction Monitoring (MRM)
- Dwell Time: 20-50 ms per transition
- Collision Energy: Optimized for each target peptide [48]
Data Processing and Statistical Analysis
Biomarker Quantification:
- Peak Integration: Manually review and integrate all MRM transitions
- Normalization: Normalize peak areas using internal standards
- Concentration Calculation: Calculate protein concentrations from peptide responses
Risk Score Calculation:
- Algorithm: Combine three protein biomarker concentrations with clinical parameters (age, HDL-cholesterol, eGFR)
- Risk Categories: Classify patients as low (<10%), moderate (10% to <20%), or high (≥20%) risk [95]
- Statistical Analysis: Assess performance using ROC curves, sensitivity, specificity, PPV, and NPV [95]
Pathway Diagrams & Research Reagents
Experimental Workflow Diagram
Biomarker Validation Pathway
Research Reagent Solutions
Table 4: Essential Research Materials for DKD Biomarker Studies
| Reagent/Instrument | Manufacturer | Specific Use Case | Key Function |
|---|---|---|---|
| MARS14 Column | Agilent Technologies | Plasma immunodepletion | Removes 14 most abundant proteins [48] |
| iTRAQ Reagents | Sciex | Discovery proteomics | Multiplexed relative protein quantitation [48] |
| Triple Quadrupole MS | Various | Targeted quantitation | MRM analysis of biomarker peptides [48] |
| Strata-X Columns | Phenomenex | Sample cleanup | Peptide desalting and concentration [48] |
| PromarkerD Test | Proteomics International | Clinical validation | Validated biomarker panel for DKD risk [95] |
This case study demonstrates a successful pipeline from mass spectrometry-based biomarker discovery to clinical validation, resulting in a commercially available test with proven utility for predicting diabetic kidney disease progression. The integration of triple-quadrupole mass spectrometry with rigorous clinical study design provides a template for future biomarker development efforts across various disease areas.
The discovery of protein biomarkers has been revolutionized by advanced mass spectrometry (MS) techniques, particularly in plasma proteomics. However, a significant bottleneck persists in translating promising biomarker candidates from discovery into validated, routine clinical tests [6]. For decades, the clinical validation and implementation of these biomarkers have been constrained by the reliance on dated triple quadrupole (QqQ) MS technology, which, while robust, lacks the speed and multiplexing capabilities required for efficient translation of large biomarker panels [6] [4]. This gap between discovery and clinical application has hindered the diagnostic and prognostic utility of protein biomarkers in precision medicine.
The emerging solution lies in a new class of hybrid high-speed mass spectrometers. Instruments like the Stellar MS integrate the robustness of traditional triple quadrupoles with the enhanced capabilities of advanced linear ion trap analyzers [6] [97]. This technological evolution creates a bridge, enabling the extremely rapid and sensitive targeted verification of thousands of peptide candidates originally identified in discovery-phase, high-resolution experiments [6]. By leveraging these hybrid systems alongside innovative quantification strategies, such as the use of 15N-labeled protein standards, researchers can now streamline the path from biomarker candidate identification to clinically actionable diagnostic tests [6] [97].
The core innovation in bridging discovery and clinical application is the hybrid mass spectrometer architecture. Unlike traditional triple quadrupoles used for targeted quantitation, these systems combine different mass analyzer technologies to overcome historical limitations.
The Stellar MS exemplifies this hybrid approach by integrating a triple quadrupole framework with an advanced linear ion trap (LIT) analyzer [6] [97]. This configuration allows the instrument to operate in highly sensitive Parallel Reaction Monitoring (PRM) mode and perform MS3 scanning, providing an additional layer of specificity for challenging clinical matrices [6]. While traditional triple quadrupoles use Multiple Reaction Monitoring (MRM) to monitor a few predefined ion transitions, the hybrid system can simultaneously target thousands of peptides with high specificity and speed, making it ideal for verifying large biomarker panels from discovery-phase data-independent acquisition (DIA) studies [6].
This architecture directly addresses the key challenge in clinical proteomics: the need for absolute quantification. The system is designed to work efficiently with stable isotope-labeled standards, particularly 15N-labeled full-length proteins, which enable precise, multiplexed quantification of candidate biomarkers in a generic, streamlined workflow [6] [97]. The system's speed allows it to maintain high reproducibility and low coefficients of variation (CV) even when targeting extensive peptide panels in complex plasma samples, thus meeting the rigorous demands of clinical assay development [6].
System Workflow Diagram
The following diagram illustrates the integrated workflow from biomarker discovery to clinical application using a hybrid MS system:
Application Data: Performance Metrics for Clinical Translation
The transition from discovery proteomics to clinically viable assays requires demonstrating robust performance metrics. The hybrid MS platform has been evaluated for its ability to target thousands of peptides originally measured on discovery-grade instruments like the Orbitrap Astral MS, achieving the sensitivity and specificity necessary for quantifying many of the top 1000 plasma proteins [6]. This performance is crucial, as plasma represents one of the most challenging biological matrices due to its extreme dynamic range of protein concentrations.
In a practical demonstration of its clinical utility, targeted assays were developed for alcohol-related liver disease (ALD) biomarkers [6] [97]. The system achieved high reproducibility and low coefficients of variation, essential parameters for clinical tests where precision is non-negotiable. The quantitative capabilities were further enhanced through the use of 15N-labeled protein standards, which facilitate absolute quantification—a mandatory requirement for clinical diagnostics [6]. This approach provides a more generic and streamlined alternative to peptide-specific stable isotope labels, simplifying the assay development process for multiple biomarker panels.
Table 1: Performance Metrics of Hybrid MS Systems in Biomarker Verification
| Performance Parameter | Capability Demonstrated | Clinical Significance |
|---|---|---|
| Throughput & Speed | Targets thousands of peptides in short LC gradients [6] | Enables high-volume verification of biomarker candidates |
| Reproducibility | Achieves low coefficients of variation (CV) [6] | Meets regulatory requirements for clinical test precision |
| Sensitivity | Sufficient for top 1000 plasma proteins [6] | Detects low-abundance, clinically relevant biomarkers |
| Specificity | Utilizes PRM and MS3 capabilities [6] | Reduces false positives in complex clinical samples |
| Quantification | Absolute quantification with 15N-labeled standards [6] [97] | Provides concentration data required for clinical decision-making |
Experimental Protocol: From Discovery Data to Targeted Clinical Assay
Protocol: Translation of DIA Data to Targeted PRM Assays Using Hybrid MS
Objective: To translate biomarker candidates identified in discovery-phase DIA experiments into targeted, quantitative assays for clinical verification using a hybrid mass spectrometer.
Materials & Reagents:
- Liquid chromatography system (nanoflow or analytical flow)
- Hybrid mass spectrometer (e.g., Stellar MS with LIT analyzer)
- 15N-labeled protein standards (for absolute quantification) [6] [97]
- Plasma samples (depleted or non-depleted)
- Digestion buffer (e.g., Tris-HCl, ammonium bicarbonate)
- Reduction and alkylation reagents (DTT, iodoacetamide)
- Trypsin (sequencing grade)
- Solid-phase extraction cartridges (e.g., C18 for desalting)
Procedure:
Sample Preparation:
- Deplete high-abundance proteins from plasma samples if necessary [6].
- Reduce disulfide bonds with 5mM DTT (30 min, 60°C).
- Alkylate with 15mM iodoacetamide (30 min, room temperature in the dark).
- Digest proteins with trypsin (1:50 enzyme-to-protein ratio) overnight at 37°C.
- Add 15N-labeled protein standards to the digested sample [6] [97].
- Desalt peptides using C18 solid-phase extraction.
LC-MS/MS Analysis:
- Separate peptides using a reversed-phase nanoLC or analytical LC gradient (e.g., 15-60 min) [6].
- Operate the hybrid MS in Parallel Reaction Monitoring (PRM) mode.
- Import the list of candidate peptides and proteins identified from prior DIA discovery data.
- For each target peptide, configure the method to isolate the precursor ion in Q1 and fragment it in the collision cell.
- Acquire full-scan MS2 spectra in the linear ion trap analyzer for all fragment ions.
- For increased specificity in complex samples, implement MS3 scanning for a subset of key biomarkers [6].
Data Processing & Quantification:
- Process PRM data using software (e.g., Skyline) to extract ion chromatograms for fragment ions of each target peptide.
- Integrate peak areas for endogenous peptides and their corresponding 15N-labeled internal standard peptides.
- Calculate the ratio of endogenous to standard peptide for absolute quantification [6] [97].
- Generate calibration curves using the stable isotope-labeled standards to determine the concentration of the biomarker in the clinical sample.
Technology Comparison Diagram
The following diagram contrasts the traditional workflow with the new hybrid MS-based approach for biomarker development:
The Scientist's Toolkit: Essential Research Reagent Solutions
The successful implementation of a biomarker verification pipeline relies on a suite of essential reagents and materials. The table below details key solutions for developing targeted clinical assays using hybrid MS systems.
Table 2: Key Research Reagent Solutions for Biomarker Verification
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| 15N-Labeled Protein Standards | Enable absolute quantification in a generic, multiplexed manner; act as internal standards for precise measurement [6] [97] | Spiked into plasma samples prior to digestion to account for sample preparation losses and ionization variability |
| Stable Isotope-Labeled (SIS) Peptides | Traditional standards for precise quantification of specific proteotypic peptides; used in MRM/PRM assays [2] | Used for assay optimization and as quantitation references for specific peptide targets |
| High-Affinity Depletion Columns | Remove high-abundance proteins (e.g., albumin, IgG) from plasma/serum to enhance detection of lower-abundance biomarkers [6] | Pre-fractionation of clinical plasma samples to deepen proteome coverage and improve assay sensitivity |
| Sequencing-Grade Trypsin | Proteolytic enzyme for specific digestion of proteins into peptides for bottom-up proteomics [2] | Standardized digestion of protein samples to generate representative peptides for LC-MS/MS analysis |
| Solid-Phase Extraction (SPE) Cartridges | Desalt and concentrate peptide mixtures after digestion, improving LC-MS performance and stability [10] | Clean-up of digested plasma samples prior to LC-MS injection to remove interfering salts and buffers |
Hybrid high-speed mass spectrometers represent a transformative advancement in clinical proteomics, effectively bridging the long-standing gap between biomarker discovery and routine clinical testing. By integrating the robustness of triple quadrupole instruments with the speed and specificity of advanced linear ion traps, platforms like the Stellar MS enable the rapid verification of large biomarker panels with the reproducibility and precision required for clinical applications [6]. The synergistic use of these hybrid systems with innovative quantification methods, such as 15N-labeled protein standards, creates a streamlined, generic pipeline for translating discovery-phase findings into clinically actionable assays [6] [97]. As these technologies continue to evolve and become more accessible, they hold the potential to significantly accelerate the adoption of protein biomarkers in precision medicine, ultimately enhancing diagnostic and prognostic capabilities across a wide spectrum of diseases.
Conclusion
Triple quadrupole mass spectrometry, particularly using SRM/MRM, remains the cornerstone for precise and reliable biomarker validation, bridging the critical gap between discovery and clinical application. Its unparalleled sensitivity, specificity, and quantitative robustness make it indispensable for developing multiplexed assays in complex matrices. While immunoassays face challenges with cross-reactivity and high-resolution platforms excel in untargeted discovery, QqQ occupies a unique, vital niche for targeted quantification. Future directions point towards the integration of QqQ with more advanced analyzers in hybrid systems, the use of heavy isotope-labeled standards for absolute quantification, and streamlined workflows that accelerate the translation of biomarker panels into clinical tools for personalized medicine, ultimately improving diagnostic and prognostic capabilities across a spectrum of diseases.
References
- 1. Triple quadrupole mass spectrometer [https://en.wikipedia.org/wiki/Triple_quadrupole_mass_spectrometer]
- 2. Triple Quadrupole Mass Spectrometer - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/triple-quadrupole-mass-spectrometer]
- 3. Current Role and Potential of Triple Quadrupole Mass ... [https://www.mdpi.com/1420-3049/29/23/5808]
- 4. Current Role and Potential of Triple Quadrupole Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11643727/]
- 5. Triple Quadrupole LC-MS/MS [https://www.shimadzu.com/an/products/liquid-chromatograph-mass-spectrometry/triple-quadrupole-lc-msms/index.html]
- 6. A Novel Hybrid High-Speed Mass Spectrometer Allows ... [https://pubmed.ncbi.nlm.nih.gov/40816420/]
- 7. SRM/MRM: Principles, Applications & Instrumentation [https://www.creative-proteomics.com/resource/srm-mrm-techniques-principles-applications-instrumentation.htm]
- 8. SRM/MRM targeted proteomics as a tool for biomarker ... [https://pubmed.ncbi.nlm.nih.gov/26472065/]
- 9. Selected reaction monitoring [https://en.wikipedia.org/wiki/Selected_reaction_monitoring]
- 10. Mass spectrometry based biomarker discovery, verification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5528535/]
- 11. Selected reaction monitoring for quantitative proteomics [https://pmc.ncbi.nlm.nih.gov/articles/PMC2583086/]
- 12. PRM vs MRM: A Comparative Guide to Targeted ... [https://www.metwarebio.com/prm-vs-mrm-targeted-ms-comparison/]
- 13. Mass spectrometry based biomarker discovery, verification ... [https://www.sciencedirect.com/science/article/pii/S1574789114000544]
- 14. Selected reaction monitoring (SRM) mass spectrometry ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4121859/]
- 15. MRMPlus: an open source quality control and assessment tool ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-015-0838-z]
- 16. Comparison of Quantitative Mass Spectrometry Platforms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6021760/]
- 17. The tar pit of the protein biomarker pipeline [https://pmc.ncbi.nlm.nih.gov/articles/PMC2957839/]
- 18. Biomarker Discovery: Types, Phases and Applications [https://lifesciences.danaher.com/us/en/library/biomarker-discovery.html]
- 19. Isobaric mass tagging and triple quadrupole mass ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0226073]
- 20. Tutorial: best practices and considerations for mass ... [https://www.nature.com/articles/s41596-021-00566-6]
- 21. Ten quick tips for biomarker discovery and validation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9371329/]
- 22. Biomarker Assay Validation by Mass Spectrometry [https://pubmed.ncbi.nlm.nih.gov/35534647/]
- 23. Harnessing Mass Spectrometry for Biomarker Discovery in ... [https://blog.crownbio.com/biomarker-and-mass-spectrometry-aml]
- 24. Modeling and systematic analysis of biomarker validation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5270363/]
- 25. A Quick Introduction to Triple-Quadrupole Mass Spectrometry [https://www.jeolusa.com/NEWS-EVENTS/Blog/a-quick-introduction-to-triple-quadrupole-mass-spectrometry]
- 26. Triple Quadrupole Mass Spectrometer (QqQ MS) [https://www.labcompare.com/Mass-Spectrometry/131-Triple-Quadrupole-Mass-Spectrometer-QqQ-MS/]
- 27. A Workflow to Perform Targeted Metabolomics at the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8377714/]
- 28. Triple quadrupole–mass spectrometry protocols for the ... [https://www.sciencedirect.com/science/article/abs/pii/S0379073820300463]
- 29. Immunoassays and Mass Spectrometry for Determination ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11046482/]
- 30. Modeling and systematic analysis of biomarker using... validation [https://bsb-eurasipjournals.springeropen.com/articles/10.1186/s13637-014-0017-y]
- 31. Proteotypic Peptides and Their Applications [https://pubmed.ncbi.nlm.nih.gov/27975286/]
- 32. The use of proteotypic peptide libraries for protein ... [https://pubmed.ncbi.nlm.nih.gov/15945033/]
- 33. Rapid Optimization of MRM-MS Instrument Parameters by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2811718/]
- 34. An extra dimension in protein tagging by quantifying ... [https://www.nature.com/articles/srep27220]
- 35. Assigning statistical significance to proteotypic peptides via ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3186061/]
- 36. Mass Spectrometry Quantitation [https://www.ionsource.com/tutorial/msquan/srm.htm]
- 37. Development and Optimization of a High Sensitivity LC-MS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7570656/]
- 38. Development and validation of a liquid chromatography- triple ... [https://pubmed.ncbi.nlm.nih.gov/37126878/]
- 39. Protein Sample Preparation for Mass Spectrometry [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/sample-preparation-mass-spectrometry.html]
- 40. Sample preparation for serum/plasma profiling and biomarker ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7094463/]
- 41. Blood Plasma and Serum Preparation [https://www.thermofisher.com/us/en/home/references/protocols/cell-and-tissue-analysis/elisa-protocol/elisa-sample-preparation-protocols/blood-plasma-serum-preparation.html]
- 42. Development and validation of a liquid chromatography ... [https://www.sciencedirect.com/science/article/pii/S0021967323002285]
- 43. Compensate for or Minimize Matrix Effects? Strategies ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7412464/]
- 44. What are Matrix Effect in Liquid Chromatography Mass ... [https://www.nebiolab.com/what-are-matrix-effect-in-liquid-chromatography-mass-spectrometry/]
- 45. Biological Matrix Effects in Quantitative Tandem Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4695332/]
- 46. Matrix Effects on Quantitation in Liquid Chromatography [https://www.chromatographyonline.com/view/matrix-effects-on-quantitation-in-liquid-chromatography-sources-and-solutions]
- 47. Recent developments in mass-spectrometry-based targeted ... [https://clinicalproteomicsjournal.biomedcentral.com/articles/10.1186/s12014-024-09452-1]
- 48. Comprehensive mass spectrometry based biomarker ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5988498/]
- 49. Recent progress in mass spectrometry-based liquid biopsy ... [https://www.sciencedirect.com/science/article/pii/S0165993625001591]
- 50. A robust multiplex immunoaffinity mass spectrometry assay ... [https://clinicalproteomicsjournal.biomedcentral.com/articles/10.1186/s12014-020-09302-w]
- 51. Urinary peptidomics and bioinformatics for the detection of ... [https://www.nature.com/articles/s41598-020-58067-7]
- 52. Mass Spectrometry for Clinical Research [https://www.thermofisher.com/us/en/home/clinical/clinical-translational-research/mass-spectrometry-applications-clinical-research/ms-clinical-research.html]
- 53. Quantitative high-throughput screening data analysis [https://pmc.ncbi.nlm.nih.gov/articles/PMC4375054/]
- 54. High Throughput Screening: Methods and Protocols [https://link.springer.com/book/10.1007/978-1-4939-3673-1]
- 55. Therapeutic drug monitoring of glycopeptide antimicrobials [https://pubmed.ncbi.nlm.nih.gov/38304144/]
- 56. Benefits of mass spectrometry in therapeutic drug monitoring [https://diagnostics.roche.com/global/en/article-listing/lab-leaders/article/benefits-mass-spectrometry-in-tdm.html]
- 57. High-throughput kinetics in drug discovery [https://www.sciencedirect.com/science/article/pii/S2472555224000327]
- 58. Optimizing LC‑MS/MS Sensitivity: Strategies to Overcome ... [https://biotech-spain.com/en/articles/optimizing-lc-ms-ms-sensitivity-strategies-to-overcome-ion-suppression-and-boost-robustness-in-bioanalysis/]
- 59. Elucidating the Role of Ion Suppression in Secondary ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10623576/]
- 60. Ion suppression correction and normalization for non- ... [https://www.nature.com/articles/s41467-025-56646-8]
- 61. Study of Metabolite Detectability in Simultaneous Profiling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12388855/]
- 62. Reduced Pressure Ionization Enhances Native Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12559447/]
- 63. Mass Resolution and Mass Accuracy: How Much Is Enough? [https://pmc.ncbi.nlm.nih.gov/articles/PMC3809961/]
- 64. Triple Quadrupole Mass Spectrometry - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/triple-quadrupole-mass-spectrometry]
- 65. Learn with Lin about LC–MS bioanalysis: part III tune ... [https://www.bioanalysis-zone.com/learn-with-lin-about-lc-ms-bioanalysis-part-iii-tune-the-triple-quadrupole-system_covance/]
- 66. Optimization of the Ion Source-Mass Spectrometry ... [https://link.springer.com/article/10.1007/s13361-016-1459-0]
- 67. Effect of Collision Energy Optimization on the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3005404/]
- 68. Data-Driven Optimization of Mass Spectrometry Methods [https://pmc.ncbi.nlm.nih.gov/articles/PMC6737531/]
- 69. Multi-Parameter Auto-Tuning Algorithm for Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10525632/]
- 70. USP Signal-to-Noise Ratios: Standards and Challenges in ... [https://www.sepscience.com/usp-signal-to-noise-ratios-standards-and-challenges-in-chromatography-11213]
- 71. Introduction to mass spectrometry data analysis [https://www.aspect-analytics.com/media-blog/introduction-to-mass-spectrometry-data-analysis]
- 72. The Role of Solvent Gradients in HPLC Analysis and How ... [https://chromtech.com/blog/the-role-of-solvent-gradients-in-hplc-analysis-and-how-asi-static-mixers-can-improve-performance/]
- 73. The Essential Guide to Optimizing Sensitivity in GC–FID ... [https://www.chromatographyonline.com/view/essential-guide-optimizing-sensitivity-gc-fid-analysis]
- 74. Single to Triple: Fundamentals and Modes of Bench-Top ... [https://www.chromatographyonline.com/view/single-to-triple-fundamentals-and-modes-of-bench-top-gas-chromatography-triple-quadrupole-mass-spectrometry-gc-ms-ms-]
- 75. Blood proteomics for quantitative biomarkers of cellular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12481993/]
- 76. Analytical Validation Considerations of Multiplex Mass- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4261948/]
- 77. Limit of Blank, Limit of Detection and Limit of Quantitation [https://pmc.ncbi.nlm.nih.gov/articles/PMC2556583/]
- 78. 9.1. Definitions and important aspects – Validation of liquid ... [https://sisu.ut.ee/lcms_method_validation/91-definitions-and-important-aspects/]
- 79. Limit of Quantitation - an overview [https://www.sciencedirect.com/topics/nursing-and-health-professions/limit-of-quantitation]
- 80. Development and Validation of a Reliable UHPLC-MS/MS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8838099/]
- 81. Advancing the sensitivity of selected reaction monitoring-based targeted quantitative proteomics - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3375056/]
- 82. A review on mass spectrometry-based quantitative ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267017301502]
- 83. Mass spectrometry-based proteomics as an emerging tool in ... [https://clinicalproteomicsjournal.biomedcentral.com/articles/10.1186/s12014-023-09424-x]
- 84. Comparison of unit resolution SRM and TOF-MS at 12,000 mass resolution for quantitative bioanalysis of 11 steroids from human plasma - PubMed [https://pubmed.ncbi.nlm.nih.gov/22409553/]
- 85. A Novel Hybrid High-Speed Mass Spectrometer Allows ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12455101/]
- 86. Mass Spectrometry for the Masses [https://www.genengnews.com/topics/omics/mass-spectrometry-for-the-masses/]
- 87. Top Mass Spectrometry Instruments Compared: Features, ... [https://www.metwarebio.com/top-mass-spectrometry-technologies-comparison/]
- 88. Quadrupole Time-of-Flight Mass Spectrometry - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC6719743/]
- 89. Orbitrap LC-MS | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/industrial/mass-spectrometry/liquid-chromatography-mass-spectrometry-lc-ms/lc-ms-systems/orbitrap-lc-ms.html]
- 90. Orbitrap [https://en.wikipedia.org/wiki/Orbitrap]
- 91. Orbitrap LC-MS Comparison Chart [https://www.thermofisher.com/us/en/home/industrial/mass-spectrometry/liquid-chromatography-mass-spectrometry-lc-ms/lc-ms-systems/orbitrap-lc-ms/orbitrap-lc-ms-comparison-chart.html]
- 92. Comparison between triple quadrupole, time of flight and ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267010013838]
- 93. Insights into serum metabolic biomarkers for early ... [https://www.aging-us.com/article/205542/text]
- 94. Serum analysis of type 2 diabetes mellitus patients with low ... [https://www.sciencedirect.com/science/article/abs/pii/S1056872725002557]
- 95. the Fremantle Diabetes Study Phase II - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11466018/]
- 96. Comprehensive mass spectrometry based biomarker ... [https://www.sciencedirect.com/science/article/pii/S2212968516300393]
- 97. A Novel Hybrid High-Speed Mass Spectrometer Allows ... [https://www.sciencedirect.com/science/article/pii/S1535947625001495]